有机化学 ›› 2026, Vol. 46 ›› Issue (4): 1529-1539.DOI: 10.6023/cjoc202512042 上一篇 下一篇
综述与进展
收稿日期:2025-12-28
修回日期:2026-01-26
发布日期:2026-03-06
通讯作者:
朱峰, 杨波
基金资助:
Yujuan Wang, Feng Zhu*( ), Bo Yang*(
), Bo Yang*( )
)
Received:2025-12-28
Revised:2026-01-26
Published:2026-03-06
Contact:
Feng Zhu, Bo Yang
Supported by:文章分享
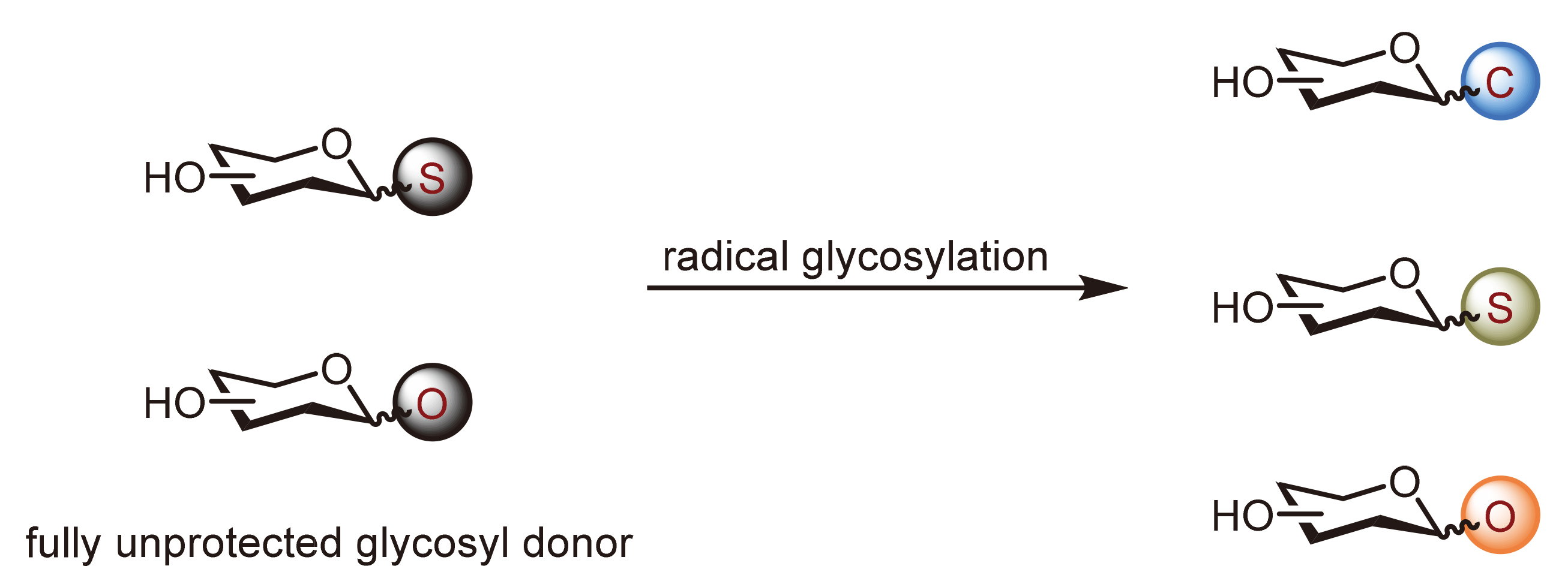
综述了糖化学前沿领域的最新研究进展, 重点聚焦于无保护糖的糖苷化反应. 糖类是重要的生物信息分子, 其合成高度依赖高效的糖苷化方法. 传统策略通常包括繁琐的保护及脱保护步骤, 进一步推动了以天然糖为原料直接进行糖苷化反应策略的发展. 借助光催化和自由基介导等新型活化方式, 这类策略可将天然无保护糖一步精准转化为复杂糖缀合物. 这种新型活化方式不仅简化了合成步骤, 提高了原子经济性, 还为糖生物学研究与糖类药物开发提供了通用平台. 系统综述了各类无保护糖基供体在构建C-糖苷, S-糖苷与O-糖苷键中的应用, 并对未来研究方向进行了展望.
王钰涓, 朱峰, 杨波. 完全未保护糖的自由基糖基化反应[J]. 有机化学, 2026, 46(4): 1529-1539.
Yujuan Wang, Feng Zhu, Bo Yang. Radical Glycosylation of Fully Unprotected Sugars[J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1529-1539.
| [1] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
| [2] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [1] | 李欢乐, 潘其, 娄绍杰, 毛羊杰, 许丹倩. 基于氢原子迁移(HAT)过程的胺类化合物选择性C—H键转化反应研究进展[J]. 有机化学, 2025, 45(9): 3213-3243. |
| [2] | 钟国, 白萌冉, 崔斌, 孙慧. 烯烃立体选择性卤胺化的研究进展[J]. 有机化学, 2025, 45(12): 4271-4289. |
| [3] | 杜佳言, 刘俊涛, 刘桂霞, 黄正. 钴催化末端烯烃区域和立体选择性异构合成反式-2-烯烃[J]. 有机化学, 2024, 44(9): 2889-2897. |
| [4] | 李平, 张寅, 杨子琪, 郝文娟, 姜波. 利用碱促进环外1,3-二羰化合物的解构反应合成腙化的1,n-二羰化合物及其生物活性检测[J]. 有机化学, 2024, 44(9): 2777-2784. |
| [5] | 王兢睿, 冯永奎, 王能中, 黄年玉, 姚辉. 钯催化立体选择性合成硝基烷类β-碳糖苷[J]. 有机化学, 2023, 43(9): 3216-3225. |
| [6] | 向勋, 何照林, 董秀琴. 钯和手性磷酸协同催化高效构建手性分子的研究进展[J]. 有机化学, 2023, 43(3): 791-808. |
| [7] | 潘康, 徐凡. 硅氨基镧化合物催化合成磷酸烯醇酯[J]. 有机化学, 2023, 43(12): 4261-4267. |
| [8] | 田冲, 孙奇, 王俊锋, 陈俏, 温志国, Maxim Borzov, 聂万丽. 卤素阴离子催化的立体可控炔烃碳硼化反应研究[J]. 有机化学, 2023, 43(1): 338-344. |
| [9] | 孙奇, 孙泽颖, 俞泽, 王光伟. 镍催化炔烃的立体选择性芳基-二氟烷基化反应[J]. 有机化学, 2022, 42(8): 2515-2520. |
| [10] | 来梦楠, 王秋圆, 华敏, 黄年玉, 姚辉. 开放体系中芳基岩藻糖/阿拉伯糖碳苷的立体选择性合成[J]. 有机化学, 2022, 42(6): 1694-1705. |
| [11] | 高娜, 初晓辉, 刘洋, 李家柱, 王进军. 焦脱镁叶绿酸的区域和立体选择性的芳(芳酰)亚甲基化及其叶绿素类二氢卟吩衍生物的合成[J]. 有机化学, 2022, 42(4): 1111-1122. |
| [12] | 李征, 谷迎春, 徐大振, 费学宁, 张磊. 有机膦催化的[4+2]环加成反应机理的密度泛函理论研究[J]. 有机化学, 2022, 42(3): 830-837. |
| [13] | 孙阳星, 李彦芝, 曹燕来, 王能中, 向少华, 刘明国, 姚辉. 官能团导向立体选择性的糖苷化反应研究进展[J]. 有机化学, 2022, 42(11): 3506-3529. |
| [14] | 平媛媛, 宋海霞, 孔望清. 镍催化烯烃的不对称还原双官能团化反应研究进展[J]. 有机化学, 2022, 42(10): 3302-3321. |
| [15] | 庞海亮, 吴冬, 阴国印. 钯催化的1,4-环己二烯的立体选择性1,3-双芳基化反应[J]. 有机化学, 2021, 41(2): 849-856. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||