化学学报 ›› 2026, Vol. 84 ›› Issue (3): 299-304.DOI: 10.6023/A25120430 上一篇 下一篇
研究论文
王怡可†, 陈丽萍†, 舒敬利†, 朱雪华*( ), 汪洋*(
), 汪洋*( )
)
投稿日期:2025-12-30
发布日期:2026-02-10
基金资助:
Wang Yike, Chen Liping, Shu Jingli, Zhu Xuehua*( ), Wang Yang*(
), Wang Yang*( )
)
Received:2025-12-30
Published:2026-02-10
Contact:
*E-mail: zhuxuehua@usts.edu.cn,
ywang@usts.edu.cn
About author:† These authors contributed equally to this work.
Supported by:文章分享
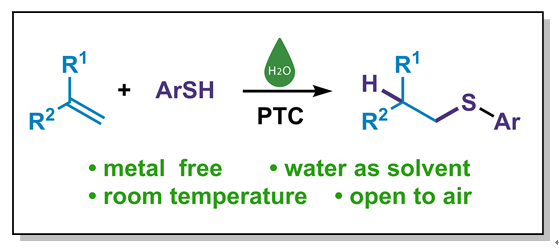
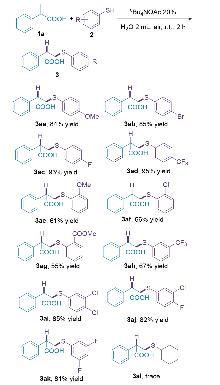
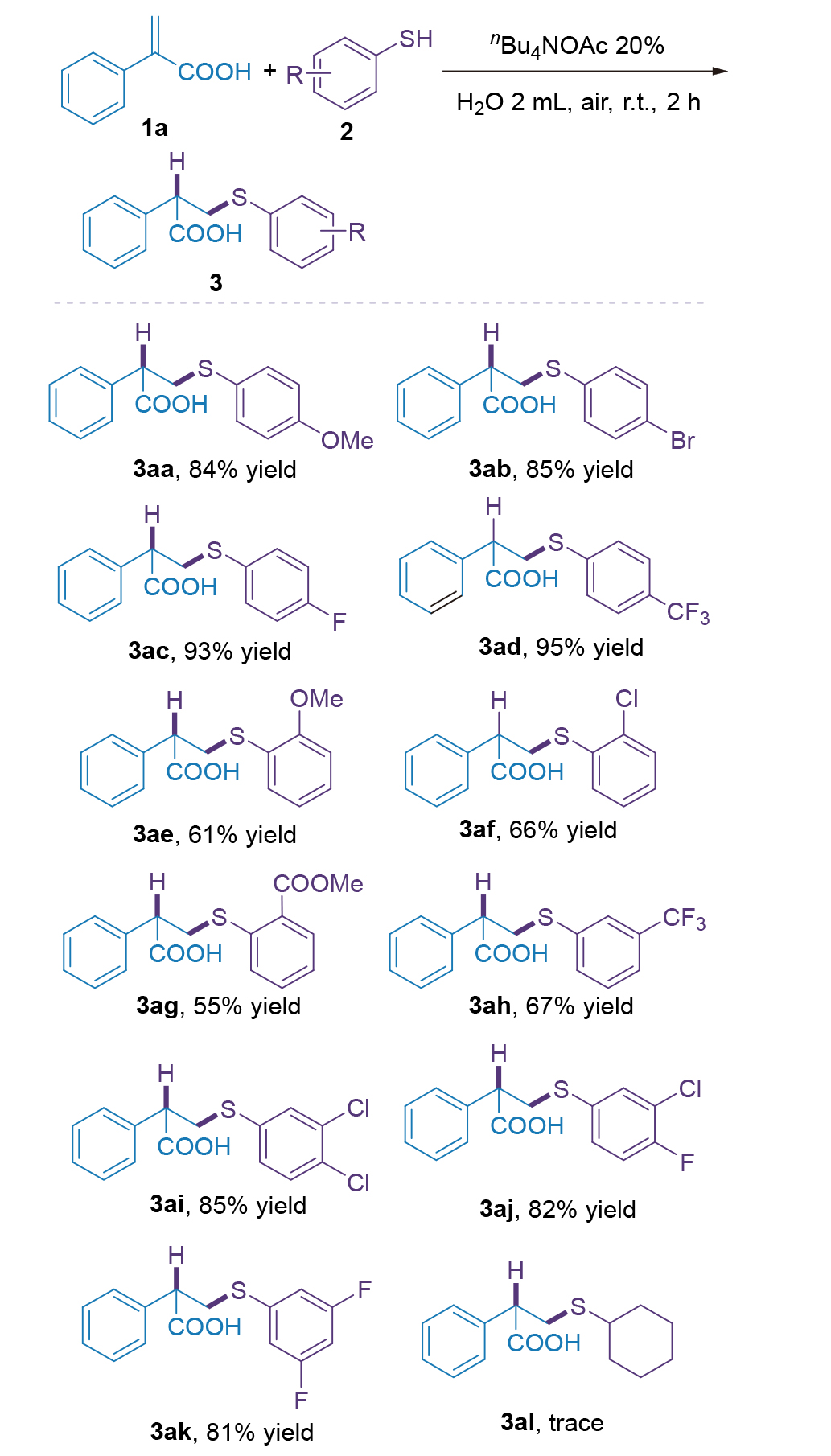
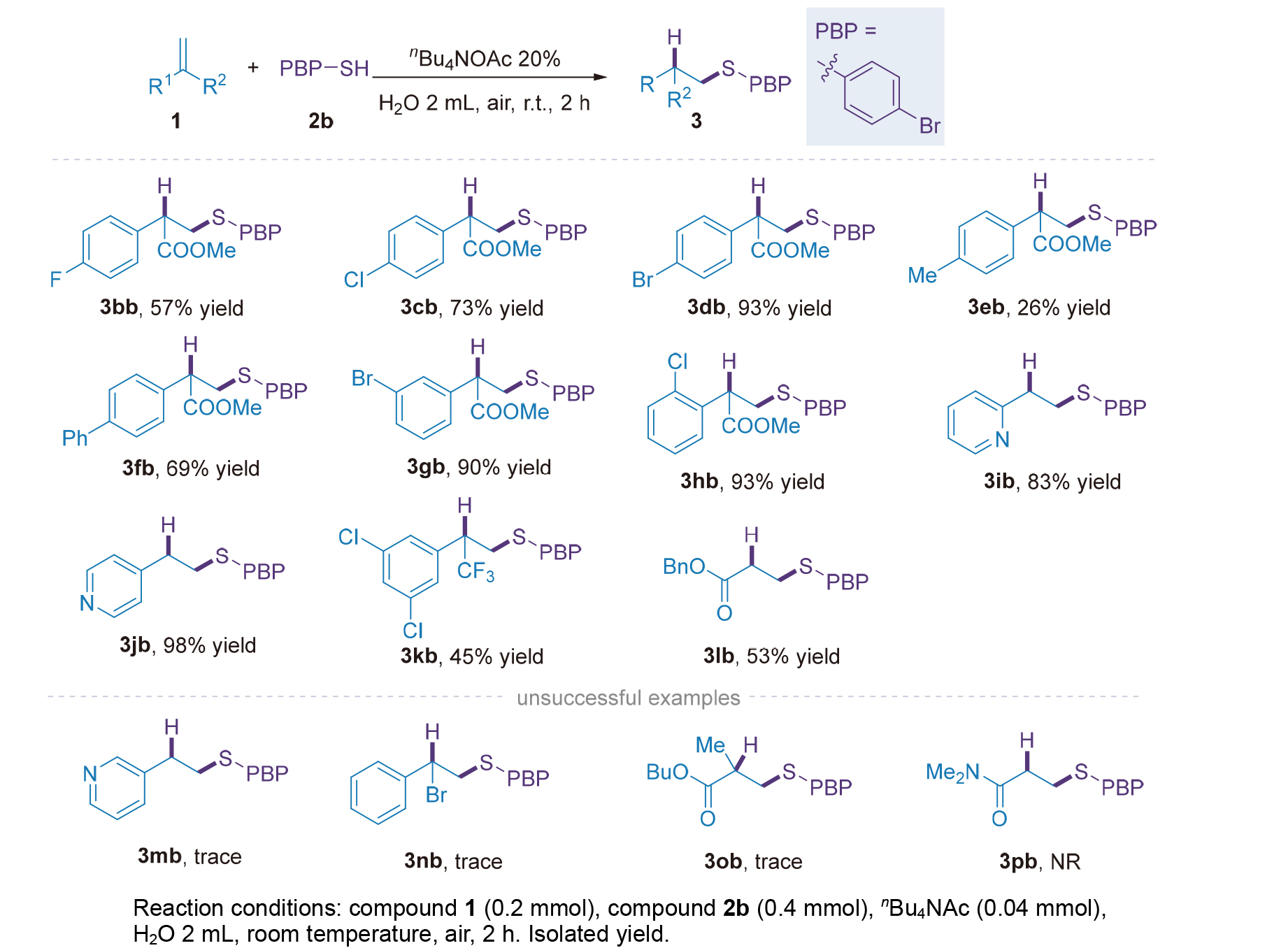
有机硫化合物是生物活性分子与功能材料的关键合成子, 高效高选择性构建C—S键为有机合成核心方向. 现有烯烃反马氏硫氢化方法常依赖昂贵催化剂、反应条件严苛, 且水相适用性差, 限制其绿色化应用. 本研究开发水相烯烃反马氏硫氢化新方法, 以四丁基醋酸铵(nBu₄NOAc)为最优相转移催化剂, 室温空气下反应2 h即可高产率获得产物, 无需金属、碱或惰性气保护. 该方法对多类取代硫酚及烯烃兼容性良好, 收率优良且卤素可保留. 克级反应收率良好, 具有一定的工业化潜力, 产物的后期拓展性强, 具备一定应用潜力. 机理推测存在氢键诱导与亲核加成两条路径, nBu₄NOAc起双重催化作用. 该方法条件温和、操作简便且环境友好, 为C—S键构建提供绿色新策略.
王怡可, 陈丽萍, 舒敬利, 朱雪华, 汪洋. 基于相转移催化的水相烯烃高效反马氏硫氢化反应[J]. 化学学报, 2026, 84(3): 299-304.
Wang Yike, Chen Liping, Shu Jingli, Zhu Xuehua, Wang Yang. Efficient Anti-Markovnikov Hydrothiolation of Alkenes via Phase-Transfer Catalysis in Aqueous Phase[J]. Acta Chimica Sinica, 2026, 84(3): 299-304.
| Entry | Variation from the standard conditions | Yieldb/% |
|---|---|---|
| 1 | none | 85 |
| 2 | nBu4NBF4 instead of nBu4NOAc | 65 |
| 3 | nBu4NPF6 instead of nBu4NOAc | 25 |
| 4 | TEBAC instead of nBu4NOAc | 55 |
| 5 | nBu4NClO4 instead of nBu4NOAc | n.r. |
| 6 | nBu4NI instead of nBu4NOAc | trace |
| 7 | nBu4NBr instead of nBu4NOAc | trace |
| 8 | nBu4NCl instead of nBu4NOAc | trace |
| 9 | 18-Crown-6 instead of nBu4NOAc | n.r. |
| 10 | KOAc instead of nBu4NOAc | 31 |
| 11 | K2CO3 instead of nBu4NOAc | n.r. |
| 12 | Na2CO3 instead of nBu4NOAc | n.r. |
| 13 | No PTC | n.r. |
| 14 | 40 ℃ | 83 |
| 15 | N2 | 85 |
| 16 | Neat | n.r. |
| Entry | Variation from the standard conditions | Yieldb/% |
|---|---|---|
| 1 | none | 85 |
| 2 | nBu4NBF4 instead of nBu4NOAc | 65 |
| 3 | nBu4NPF6 instead of nBu4NOAc | 25 |
| 4 | TEBAC instead of nBu4NOAc | 55 |
| 5 | nBu4NClO4 instead of nBu4NOAc | n.r. |
| 6 | nBu4NI instead of nBu4NOAc | trace |
| 7 | nBu4NBr instead of nBu4NOAc | trace |
| 8 | nBu4NCl instead of nBu4NOAc | trace |
| 9 | 18-Crown-6 instead of nBu4NOAc | n.r. |
| 10 | KOAc instead of nBu4NOAc | 31 |
| 11 | K2CO3 instead of nBu4NOAc | n.r. |
| 12 | Na2CO3 instead of nBu4NOAc | n.r. |
| 13 | No PTC | n.r. |
| 14 | 40 ℃ | 83 |
| 15 | N2 | 85 |
| 16 | Neat | n.r. |
| [1] |
doi: 10.1039/C8NP00093J |
| [2] |
doi: 10.1021/acs.chemrev.6b00697 pmid: 28418240 |
| [3] |
doi: 10.1039/b609523m |
| [4] |
doi: 10.2174/1568026615666150915111741 |
| [5] |
|
| [6] |
doi: 10.1016/j.ejmech.2018.11.017 |
| [7] |
|
| [8] |
doi: 10.1002/advs.v9.4 |
| [9] |
doi: 10.1016/j.mattod.2023.02.027 |
| [40] |
doi: 10.1039/D1GC00098E |
| [41] |
doi: 10.1038/s41467-018-03698-8 |
| [42] |
doi: 10.1021/jacs.0c11815 |
| [10] |
doi: 10.1039/D1DT04371D |
| [11] |
doi: 10.1038/s41570-023-00505-x |
| [12] |
doi: 10.1039/C8GC01337C |
| [13] |
doi: 10.1021/acs.orglett.1c02738 |
| [14] |
|
| [15] |
doi: 10.1021/acscatal.8b04633 |
| [16] |
doi: 10.1016/j.tet.2022.132711 |
| [17] |
doi: 10.1039/d1ob01091c pmid: 34333579 |
| [18] |
doi: 10.1021/acssuschemeng.1c07636 |
| [19] |
doi: 10.1002/ajoc.201800639 |
| [20] |
|
| [21] |
doi: 10.1021/jo3020825 |
| [22] |
|
| [23] |
doi: 10.1038/s41467-024-52249-x |
| [24] |
doi: 10.1002/anie.v54.3 |
| [25] |
doi: 10.1021/acs.joc.3c02722 pmid: 38564378 |
| [26] |
doi: 10.1016/S0040-4020(03)00289-8 |
| [27] |
doi: 10.1002/ajoc.v7.9 |
| [28] |
doi: 10.1021/acsomega.9b01968 pmid: 31552354 |
| [29] |
doi: 10.1002/adsc.v365.24 |
| [30] |
doi: 10.1021/ja202292q pmid: 21728292 |
| [31] |
doi: 10.1002/asia.v15.6 |
| [32] |
doi: 10.1002/cptc.v2.10 |
| [33] |
|
| [34] |
doi: 10.6023/cjoc202207032 |
|
(王维, 张哲宇, 张雪, 于海丰, 罗辉, 霍东月, 徐玉澎, 赵晓波, 有机化学, 2023, 43, 742.)
|
|
| [35] |
doi: 10.6023/cjoc201904002 |
|
(胡小宇, 于海丰, 王文举, 姜思傲, 刘奇, 何洁, 有机化学, 2019, 39, 3183.)
doi: 10.6023/cjoc201904002 |
|
| [36] |
doi: 10.6023/cjoc201709021 |
|
(王伟, 吕梦娇, 赵利霞, 张娅玲, 李本浩, 李宝林, 有机化学, 2018, 38, 883.)
|
|
| [37] |
doi: 10.6023/A24110332 |
|
(杨雪, 刘妍伶, 陈霞, 周晓玉, 王爱玲, 刘海龙, 化学学报, 2025, 83, 354.)
doi: 10.6023/A24110332 |
|
| [38] |
doi: 10.1039/C8SC04271C |
| [39] |
doi: 10.1039/D4SC02681K |
| [1] | 赵馨雨, 韩燕楠, 徐吉磊, 安庆大, 肖作毅, 苏鑫, 黄家辉. 负载型离子液体催化生物基多元酸酯的制备[J]. 化学学报, 2026, 84(3): 341-352. |
| [2] | 刘雨涵, 高盼. 使用机械化学生成的钙基重格氏试剂(R-CaX)对有机卤化物进行直接硼化[J]. 化学学报, 2024, 82(11): 1114-1119. |
| [3] | 刘坜, 郑刚, 范国强, 杜洪光, 谭嘉靖. 4-酰基/氨基羰基/烷氧羰基取代汉斯酯参与的有机反应研究进展[J]. 化学学报, 2023, 81(6): 657-668. |
| [4] | 汤乔伟, 蔡小青, 殷大鹏, 孔华庭, 张祥志, 张继超, 闫庆龙, 诸颖, 樊春海. 基于点击化学的同步辐射X射线成像标签★[J]. 化学学报, 2023, 81(5): 441-444. |
| [5] | 李畅, 郑振东, 郑江南, 田瑞军. 基于可断裂双功能探针的糖蛋白分析★[J]. 化学学报, 2023, 81(12): 1673-1680. |
| [6] | 李靖鹏, 杨棋, 张周, 曾贵云, 刘腾, 黄超. 多组分连续流动高选择性合成(Z)-N-乙烯基环N,O-缩醛衍生物[J]. 化学学报, 2022, 80(11): 1463-1468. |
| [7] | 魏哲宇, 常亚林, 余焓, 韩生, 魏永革. Anderson型杂多酸作为催化剂在有机合成中的应用[J]. 化学学报, 2020, 78(8): 725-732. |
| [8] | 董奎, 刘强, 吴骊珠. 放氢交叉偶联反应[J]. 化学学报, 2020, 78(4): 299-310. |
| [9] | 叶文波, 晏子聪, 万常峰, 侯豪情, 汪志勇. 一种新的肉桂酸类化合物的脱羧/甲基化反应[J]. 化学学报, 2018, 76(2): 99-102. |
| [10] | 裴朋昆, 张凡, 易红, 雷爱文. 可见光促进的苄位Csp3-H键活化官能团化反应[J]. 化学学报, 2017, 75(1): 15-21. |
| [11] | 陆庆全, 易红, 雷爱文. 自然氧化偶联及其在碳氢功能化反应中的应用[J]. 化学学报, 2015, 73(12): 1245-1249. |
| [12] | 张丽芳, 赵杰, 王勇. 新型三唑桥联杂化复式环糊精手性固定相的构筑及性能研究[J]. 化学学报, 2015, 73(11): 1182-1188. |
| [13] | 侯占要, 胡敏标, 王维. 多金属氧簇与倍半硅氧烷簇构筑的簇-簇杂化分子的合成及自组装结构[J]. 化学学报, 2014, 72(1): 61-68. |
| [14] | 于凤丽, 王睿. 有机-无机型杂多酸相转移催化氧化脱硫性能研究[J]. 化学学报, 2014, 72(1): 105-113. |
| [15] | 来常伟, 孙莹, 杨洪, 张雪勤, 林保平. 通过“点击化学”对石墨烯和氧化石墨烯进行功能化改性[J]. 化学学报, 2013, 71(9): 1201-1224. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||