化学学报 ›› 2026, Vol. 84 ›› Issue (3): 293-298.DOI: 10.6023/A25120425 上一篇 下一篇
研究论文
赵红艳a, Ponnam, Ponnama, 李亚彤b, 李明珠b, 杨晶雯b, 陈景超a,b,*( ), 樊保敏a,b,*(
), 樊保敏a,b,*( )
)
投稿日期:2025-12-29
发布日期:2026-02-24
基金资助:
Zhao Hongyana, Ponnam Devendara, Li Yatongb, Li Mingzhub, Yang Jingwenb, Chen Jingchaoa,b,*( ), Fan Baomina,b,*(
), Fan Baomina,b,*( )
)
Received:2025-12-29
Published:2026-02-24
Contact:
*E-mail: chenjingchao84@163.com;
adams.bmf@hotmail.com
Supported by:文章分享
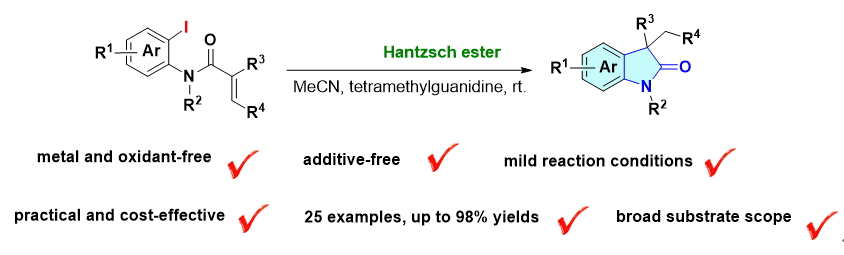
3,3-二烷基-2-吲哚酮骨架是广泛存在于天然产物和生物活性分子中的关键结构单元. 然而, 其传统合成方法通常依赖于卤代苯基丙烯酰胺的反应, 且需要使用贵金属或化学计量的有毒自由基引发剂. 本工作报道了一种无金属光化学合成3,3-二取代-2-吲哚酮的策略. 该方法以2-碘-N-甲基甲基丙烯酰胺为底物, 在可见光照射下, 利用廉价易得的汉斯酯同时作为化学计量的光还原剂和氢原子供体, 经分子内自由基环化反应高效构建目标产物. 该反应条件温和, 底物适用性广, 共合成了25个2-吲哚酮衍生物, 产率高达98%. 机理研究表明, 反应经由光诱导电子转移生成芳基自由基中间体, 随后发生5-exo-trig环化及氢原子转移, 最终得到3,3-二取代-2-吲哚酮.
赵红艳, Ponnam, Ponnam, 李亚彤, 李明珠, 杨晶雯, 陈景超, 樊保敏. 汉斯酯为光还原剂与氢源的2-吲哚酮光化学合成[J]. 化学学报, 2026, 84(3): 293-298.
Zhao Hongyan, Ponnam Devendar, Li Yatong, Li Mingzhu, Yang Jingwen, Chen Jingchao, Fan Baomin. Hantzsch Ester Serves as a Dual-function Photoreductant and Hydrogen Donor for the Metal-free Radical Cyclization Synthesis of Oxindoles[J]. Acta Chimica Sinica, 2026, 84(3): 293-298.
| Entry | Variation from the standard conditions | Yield/% |
|---|---|---|
| 1a | None | 98 |
| 2 | 5 mol% of fac-Ir(ppy)3 instead of HEH | 88 |
| 3 | 5 mol% of 4CzIPN instead of HEH | 41 |
| 4 | K2CO3/DABCO/TEA/DIPEA/Cs2CO3 (2.0 equiv.) instead of TMG | 14/15/9/5/59 |
| 5 | 1.0 equiv. of HEH used | 78 |
| 6 | 1,4-dioxane/toluene/DCM/DMSO instead of MeCN | 18/28/71/61 |
| 7 | 50 W Purple LED instead of 20 W Blue LED | 93 |
| 8 | 10 W Green LED instead of 20 W Blue LED | 5 |
| 9 | Without HEH | NR |
| 10 | Without TMG | NR |
| 11 | In dark | NR |
| 12 | In air | 36 |
| 13 | N-(2-bromophenyl)-N-methylmethacrylamide instead of 1a | 60 |
| 14 | N-(2-chlorophenyl)-N-methylmethacrylamide instead of 1a | 12 |
| Entry | Variation from the standard conditions | Yield/% |
|---|---|---|
| 1a | None | 98 |
| 2 | 5 mol% of fac-Ir(ppy)3 instead of HEH | 88 |
| 3 | 5 mol% of 4CzIPN instead of HEH | 41 |
| 4 | K2CO3/DABCO/TEA/DIPEA/Cs2CO3 (2.0 equiv.) instead of TMG | 14/15/9/5/59 |
| 5 | 1.0 equiv. of HEH used | 78 |
| 6 | 1,4-dioxane/toluene/DCM/DMSO instead of MeCN | 18/28/71/61 |
| 7 | 50 W Purple LED instead of 20 W Blue LED | 93 |
| 8 | 10 W Green LED instead of 20 W Blue LED | 5 |
| 9 | Without HEH | NR |
| 10 | Without TMG | NR |
| 11 | In dark | NR |
| 12 | In air | 36 |
| 13 | N-(2-bromophenyl)-N-methylmethacrylamide instead of 1a | 60 |
| 14 | N-(2-chlorophenyl)-N-methylmethacrylamide instead of 1a | 12 |
| [1] |
(a)
doi: 10.1021/cr300135y pmid: 31330449 |
|
(b)
doi: 10.1039/D5OB01274K pmid: 31330449 |
|
|
(c)
doi: 10.1021/jm201243p pmid: 31330449 |
|
|
(d)
doi: S0223-5234(18)30709-8 pmid: 31330449 |
|
|
(e)
doi: S0223-5234(19)30664-6 pmid: 31330449 |
|
| [2] |
(a)
doi: 10.1002/adsc.v359.11 pmid: 26419598 |
|
(b)
doi: 10.1039/D3RA02217J pmid: 26419598 |
|
|
(c)
doi: 10.1016/j.ejmech.2016.08.011 pmid: 26419598 |
|
|
(d)
doi: 10.1007/s11030-015-9629-8 pmid: 26419598 |
|
|
(e)
doi: 10.1016/j.tet.2014.08.005 pmid: 26419598 |
|
|
(f)
pmid: 26419598 |
|
|
(肖锋, 罗宇, 吕伟, 汤杰, 有机化学, 2009, 29, 459).
pmid: 26419598 |
|
| [3] |
(a)
doi: 10.1016/j.biopha.2021.111842 pmid: 34174506 |
|
(b)
pmid: 34174506 |
|
|
(c)
doi: 10.1039/D3QO00671A pmid: 34174506 |
|
|
(d)
doi: 10.1002/adsc.v352:9 pmid: 34174506 |
|
| [4] |
(a)
doi: 10.1021/acs.accounts.8b00097 pmid: 22899437 |
|
(b)
doi: 10.1039/c2cs35100e pmid: 22899437 |
|
|
(c)
doi: 10.6023/cjoc201908034 pmid: 22899437 |
|
|
(罗京华, 耿玮笙, 曹仕轩, 贺峥杰, 有机化学, 2020, 40, 40).
doi: 10.6023/cjoc201908034 pmid: 22899437 |
|
|
(d)
doi: 10.6023/A13121253 pmid: 22899437 |
|
|
(谢明胜, 武晓霞, 王刚, 林丽丽, 冯小明, 化学学报, 2014, 72, 856).
doi: 10.6023/A13121253 pmid: 22899437 |
|
|
pmid: 22899437 |
|
|
(f)
doi: 10.1039/D2OB00019A pmid: 22899437 |
|
| [5] |
(a)
doi: 10.1021/acs.accounts.0c00297 |
|
(b)
|
|
|
(c)
doi: 10.1055/a-2535-0823 |
|
|
(d)
|
|
|
(e)
doi: 10.1016/S0040-4039(01)80600-6 |
|
|
(f)
|
|
|
(g)
|
|
| [6] |
(a)
doi: 10.1016/S0040-4020(01)00634-2 |
|
(b)
doi: 10.1016/j.tet.2006.09.049 |
|
| [7] |
doi: 10.1016/0040-4039(94)02398-U |
| [8] |
(a)
doi: 10.1039/C5CC00072F |
|
(b)
doi: 10.1002/adsc.v363.18 |
|
|
(c)
doi: 10.1002/cjoc.v28:9 |
|
| [9] |
(a)
doi: 10.1021/ol301704z |
|
(b)
doi: 10.1002/cjoc.v42.11 |
|
| [10] |
(a)
doi: 10.1039/c1cs15268h pmid: 22282764 |
|
(b)
doi: 10.1021/ar7001864 pmid: 22282764 |
|
|
(c)
doi: 10.1002/asia.v2:7 pmid: 22282764 |
|
|
(d)
pmid: 22282764 |
|
|
(黄新炜, 刘建利, 有机化学, 2013, 33, 1960).
pmid: 22282764 |
|
|
(e)
doi: 10.6023/A23040118 pmid: 22282764 |
|
|
(刘坜, 郑刚, 范国强, 杜洪光, 谭嘉靖, 化学学报, 2023, 81, 657).
doi: 10.6023/A23040118 pmid: 22282764 |
|
| [11] |
(a)
doi: 10.1039/C9OB01289C |
|
(b)
|
|
|
(c)
doi: 10.1055/s-0036-1588129 |
|
| [12] |
(a)
doi: 10.1021/jacs.5c10198 |
|
(b)
doi: 10.1021/acscatal.6b02420 |
|
|
(c)
doi: 10.1021/acscatal.5c01060 |
|
|
(d)
doi: 10.1021/acs.orglett.9b04632 |
|
|
(e)
doi: 10.1021/acs.joc.3c02320 |
|
|
(f)
doi: 10.1002/adsc.v358.1 |
|
|
(g)
|
|
|
(h)
|
| [1] | 王禾林, 曾庆乐. 氧膦对吲哚炔串联加成和环化合成二芳基膦酰基吲哚并[1,2-a]喹啉[J]. 化学学报, 2026, 84(2): 189-195. |
| [2] | 汪晨程, 欧阳班来, 钟品勇, 张莺瑢, 刘晋彪. 光催化2-氨基苯甲醇与异硫氰酸酯脱硫环化合成2-氨基-1,3-苯并噁嗪[J]. 化学学报, 2026, 84(1): 1-7. |
| [3] | 李文静, 杨黎燕, 关丽, 张雪娇, 尤静, 沈思语, 赵钰琦, 段琛. 可见光/硫酚催化烯烃C=C双键的氧化裂解反应[J]. 化学学报, 2025, 83(6): 596-601. |
| [4] | 雷平, 苏秦, 王栋, Shahid Ali Khan, 阿布拉江•克依木. 无催化剂光化学合成吲哚酮: 电子供体-受体复合物介导的N-芳基丙烯酰胺环化反应[J]. 化学学报, 2025, 83(11): 1379-1385. |
| [5] | 苏秦, 雷平, 王栋, Shahid Ali Khan, 阿布拉江•克依木. 可见光诱导的α-三氟甲基烯烃与芳香酸酐的脱氟酰化反应[J]. 化学学报, 2025, 83(11): 1372-1378. |
| [6] | 李康葵, 龙先扬, 黄岳, 祝诗发. 可见光介导炔烃的自由基1,2-官能团化反应新进展[J]. 化学学报, 2024, 82(6): 658-676. |
| [7] | 黄涎廷, 韩洪亮, 肖婧, 王帆, 柳忠全. I2O5/KSCN介导的炔烃碘硫氰化反应[J]. 化学学报, 2024, 82(1): 5-8. |
| [8] | 杜思南, 赵丽曼, 张泽新, 陈国颂. 甘露糖修饰的微马达的制备及其免疫功能初探★[J]. 化学学报, 2023, 81(7): 741-748. |
| [9] | 刘坜, 郑刚, 范国强, 杜洪光, 谭嘉靖. 4-酰基/氨基羰基/烷氧羰基取代汉斯酯参与的有机反应研究进展[J]. 化学学报, 2023, 81(6): 657-668. |
| [10] | 李飞, 丁汇丽, 李超忠. 基于氟仿衍生的三氟甲基硼络合物参与的烯烃氢三氟甲基化反应[J]. 化学学报, 2023, 81(6): 577-581. |
| [11] | 徐袁利, 潘辉, 杨义, 左智伟. 连续流条件下蒽-铈协同催化的苄位碳氢键选择性氧化反应★[J]. 化学学报, 2023, 81(5): 435-440. |
| [12] | 赵亚婷, 刘帆, 汪秋安, 夏吾炯. 可见光促进(氮杂)芳香胺与重氮乙酸乙酯的N-烷基化反应[J]. 化学学报, 2023, 81(2): 111-115. |
| [13] | 解众舒, 薛中鑫, 许子文, 李倩, 王洪宇, 李维实. 石墨相氮化碳的共轭交联修饰及其对可见光催化产氢性能的影响[J]. 化学学报, 2022, 80(9): 1231-1237. |
| [14] | 赵杰, 王治文, 李华清, 艾琦, 蔡培庆, 司俊杰, 姚鑫, 胡晓光, 刘祖刚. 基于苯并[b]萘并[1,2-d]噻吩的可见光驱动光环化荧光turn-on型二芳基乙烯的合成与性能研究[J]. 化学学报, 2022, 80(9): 1223-1230. |
| [15] | 杨民, 叶柏柏, 陈健强, 吴劼. 可见光催化烷基磺酰自由基启动芳酰肼的烷基磺酰化反应[J]. 化学学报, 2022, 80(1): 11-15. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||