有机化学 ›› 2026, Vol. 46 ›› Issue (3): 859-865.DOI: 10.6023/cjoc202509036 上一篇 下一篇
研究论文
收稿日期:2025-09-28
修回日期:2025-12-16
发布日期:2026-01-15
基金资助:
Xiaoxiang Xia,b, Ming Gaob, Libiao Hana,b,*( )
)
Received:2025-09-28
Revised:2025-12-16
Published:2026-01-15
Contact:
*E-mail: Supported by:文章分享
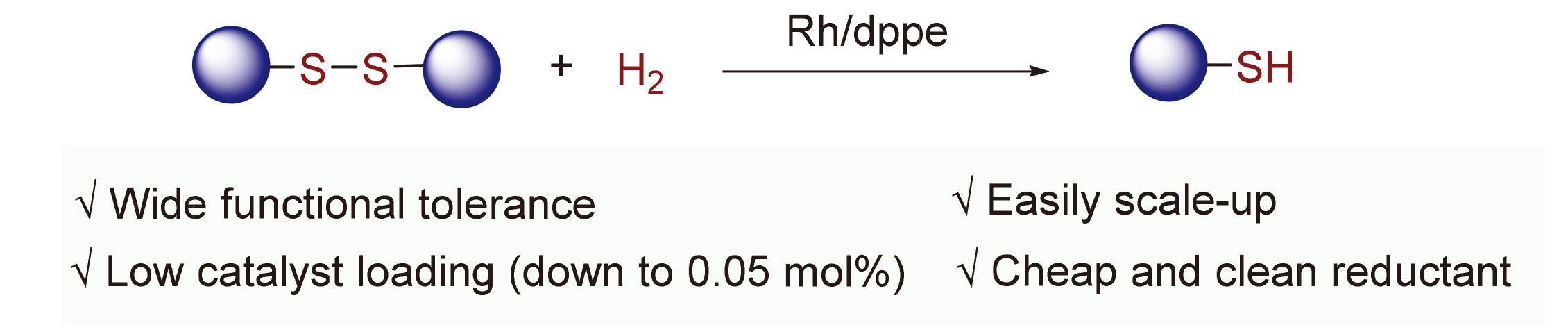
有机硫酚/醇是构建含硫化合物的合成砌块, 在有机合成化学中占有重要的地位. 报道了铑催化氢气还原二硫醚类化合物制备硫酚/醇的新方法. 该反应底物适用范围广, 并展现出良好的官能团耐受性, 多种芳基或烷基二硫醚类化物均能以良好至优异的产率被还原为相应硫酚/醇. 此外, 该方法还可以放大至克级反应规模, 铑催化剂负载量可低至0.05 mol%. 最后, 所报道的铑催化体系可循环套用三次, 其催化活性不衰减. 上述研究表明, 这种铑催化还原二硫醚类化合物的方法具有潜在的工业应用价值.
奚晓翔, 高明, 韩立彪. 铑催化氢气还原RS—SR到硫酚/醇[J]. 有机化学, 2026, 46(3): 859-865.
Xiaoxiang Xi, Ming Gao, Libiao Han. Rhodium-Catalyzed Reduction of RS—SR to Thiophenols/Thiols with H2[J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 859-865.
| Entry | Ligand (mol%) | Cat. (mol%) | Solvent | Yieldb/% |
|---|---|---|---|---|
| 1 | — | Rh-1 (5.0) | PhMe | 84 |
| 2 | — | Pt(PPh3)4 (5.0) | PhMe | N.R. |
| 3 | — | Pd(PPh3)4 (5.0) | PhMe | 33 |
| 4 | — | Ni(PPh3)4 (5.0) | PhMe | 8 |
| 5 | — | Rh-1 (1) | PhMe | 79 |
| 6c | — | Rh-1 (1) | PhMe | 75 |
| 7d | — | Rh-1 (1) | PhMe | 70 |
| 8e | — | Rh-1 (1) | PhMe | 76 |
| 9 | — | Rh-1 (0.1) | PhMe | 20 |
| 10 | PPh3 (0.1) | Rh-1 (0.1) | PhMe | 30 |
| 11 | PPh3 (0.3) | Rh-1 (0.1) | PhMe | 30 |
| 12 | — | Rh-2 (0.05) | PhMe | 6 |
| 13 | PPh3 (0.15) | Rh-2 (0.05) | PhMe | 8 |
| 14 | dppm (0.15) | Rh-2 (0.05) | PhMe | 84 |
| 15 | dppe (0.15) | Rh-2 (0.05) | PhMe | 99 (93f) |
| 16 | dppf (0.15) | Rh-2 (0.05) | PhMe | 23 |
| 17 | dpph (0.15) | Rh-2 (0.05) | PhMe | 60 |
| 18 | PCy3 (0.15) | Rh-2 (0.05) | PhMe | 30 |
| 19g | dppe (0.15) | Rh-2 (0.05) | PhMe | 90 |
| 20 | dppe (0.15) | Ru(OAc)3 (0.05) | PhMe | 82 |
| 21 | dppe (0.15) | RuCl3 (0.05) | PhMe | 80 |
| 22 | dppe (0.15) | Rh-2 (0.05) | THF | 97 |
| 23 | dppe (0.15) | Rh-2 (0.05) | DMF | 95 |
| 24 | dppe (0.15) | Rh-2 (0.05) | H2O | 70 |
| 25 | dppe (0.15) | Rh-2 (0.05) | PhC2H5 | 34 |
| 26 | dppe (0.15) | Rh-2 (0.05) | PhCN | 88 |
| 27 | dppe (0.15) | Rh-2 (0.05) | p-Xylene | 97 |
| 28 | dppe (0.15) | Rh-2 (0.05) | PhCl | 71 |
| 29h | dppe (0.15) | Rh-2 (0.05) | PhMe | 98 |
| 30 | dppe (0.15) | — | PhMe | N.R. |
| Entry | Ligand (mol%) | Cat. (mol%) | Solvent | Yieldb/% |
|---|---|---|---|---|
| 1 | — | Rh-1 (5.0) | PhMe | 84 |
| 2 | — | Pt(PPh3)4 (5.0) | PhMe | N.R. |
| 3 | — | Pd(PPh3)4 (5.0) | PhMe | 33 |
| 4 | — | Ni(PPh3)4 (5.0) | PhMe | 8 |
| 5 | — | Rh-1 (1) | PhMe | 79 |
| 6c | — | Rh-1 (1) | PhMe | 75 |
| 7d | — | Rh-1 (1) | PhMe | 70 |
| 8e | — | Rh-1 (1) | PhMe | 76 |
| 9 | — | Rh-1 (0.1) | PhMe | 20 |
| 10 | PPh3 (0.1) | Rh-1 (0.1) | PhMe | 30 |
| 11 | PPh3 (0.3) | Rh-1 (0.1) | PhMe | 30 |
| 12 | — | Rh-2 (0.05) | PhMe | 6 |
| 13 | PPh3 (0.15) | Rh-2 (0.05) | PhMe | 8 |
| 14 | dppm (0.15) | Rh-2 (0.05) | PhMe | 84 |
| 15 | dppe (0.15) | Rh-2 (0.05) | PhMe | 99 (93f) |
| 16 | dppf (0.15) | Rh-2 (0.05) | PhMe | 23 |
| 17 | dpph (0.15) | Rh-2 (0.05) | PhMe | 60 |
| 18 | PCy3 (0.15) | Rh-2 (0.05) | PhMe | 30 |
| 19g | dppe (0.15) | Rh-2 (0.05) | PhMe | 90 |
| 20 | dppe (0.15) | Ru(OAc)3 (0.05) | PhMe | 82 |
| 21 | dppe (0.15) | RuCl3 (0.05) | PhMe | 80 |
| 22 | dppe (0.15) | Rh-2 (0.05) | THF | 97 |
| 23 | dppe (0.15) | Rh-2 (0.05) | DMF | 95 |
| 24 | dppe (0.15) | Rh-2 (0.05) | H2O | 70 |
| 25 | dppe (0.15) | Rh-2 (0.05) | PhC2H5 | 34 |
| 26 | dppe (0.15) | Rh-2 (0.05) | PhCN | 88 |
| 27 | dppe (0.15) | Rh-2 (0.05) | p-Xylene | 97 |
| 28 | dppe (0.15) | Rh-2 (0.05) | PhCl | 71 |
| 29h | dppe (0.15) | Rh-2 (0.05) | PhMe | 98 |
| 30 | dppe (0.15) | — | PhMe | N.R. |
| [1] |
(a)
doi: 10.1007/s11178-005-0220-0 |
|
(b)
doi: 10.1016/S0040-4039(99)00408-6 |
|
|
(c)
doi: 10.1016/S0040-4039(00)77418-1 |
|
|
(d)
doi: 10.1016/S0040-4039(00)85691-9 |
|
|
(e)
doi: 10.3390/ma17061343 |
|
| [2] |
Our recently work on the synthesis of organophosphorus: (a)
doi: 10.1039/D3GC03071G pmid: 38602481 |
|
(b)
doi: 10.1039/D4GC04409F pmid: 38602481 |
|
|
(c)
doi: 10.1021/acs.orglett.4c00820 pmid: 38602481 |
|
|
(d)
doi: 10.1021/acs.joc.3c02678 pmid: 38602481 |
|
|
(e)
doi: 10.1021/acs.joc.3c02398 pmid: 38602481 |
|
| [3] |
doi: 10.6023/cjoc202310035 |
|
(梁家仪, 杨雅淇, 刘建平, 徐清, 韩立彪, 有机化学, 2024, 44, 1658.)
doi: 10.6023/cjoc202310035 |
|
| [4] |
(a)
pmid: 20419220 |
|
(b)
doi: 10.1002/med.2018.38.issue-4 pmid: 20419220 |
|
|
(c)
doi: 10.2174/138920309789630534 pmid: 20419220 |
|
|
(d)
doi: 10.1039/b907301a pmid: 20419220 |
|
|
(e)
doi: 10.2174/1568026615666150915111741 pmid: 20419220 |
|
| [5] |
Examples of thiophenol conversion to thioether: (a)
doi: 10.1016/j.jcat.2023.07.001 pmid: 36877862 |
|
(b)
doi: 10.1039/D3SC02440G pmid: 36877862 |
|
|
(c)
doi: 10.1039/D3QO00744H pmid: 36877862 |
|
|
(d)
doi: 10.1021/acs.orglett.3c00218 pmid: 36877862 |
|
|
(e)
doi: 10.1021/acs.joc.2c00866 pmid: 36877862 |
|
| [6] |
Examples of thiophenol conversion to sulfoxides: (a)
doi: 10.1039/C9GC00222G |
|
(b)
doi: 10.1021/acs.orglett.2c04238 |
|
|
(c)
doi: 10.1021/acs.orglett.7b03901 |
|
| [7] |
doi: 10.1002/adsc.v358.1 |
| [8] |
Examples of thiophenol conversion to sulfoxides: (a)
doi: 10.1021/jacs.9b06126 |
|
(b)
doi: 10.1002/anie.v59.52 |
|
|
(c)
doi: 10.1021/acssuschemeng.4c03951 |
|
| [9] |
doi: 10.1007/s11178-006-0009-9 |
| [10] |
doi: 10.1021/ja01304a031 |
| [11] |
|
| [12] |
Example of Matal promote reductive cleavage of the S—S:
|
|
(b)
doi: 10.1007/s10989-005-8113-1 |
|
|
(c)
doi: 10.1080/00397919708006063 |
|
| [13] |
Selected example of reductive cleavage of the S—S with hydrides: (a)
|
|
(b)
doi: 10.1021/jo00214a009 |
|
|
(c)
doi: 10.1080/00397918608059663 |
|
|
(d)
doi: 10.1139/v07-142 |
|
| [14] |
doi: 10.1080/00397918808064014 |
| [15] |
(a)
doi: 10.1021/ja00826a021 |
|
(b)
doi: 10.1021/jo00907a016 |
|
| [16] |
doi: 10.31635/ccschem.025.202505568 |
| [17] |
(a)
doi: 10.1006/jcat.1993.1321 |
|
(b)
doi: 10.1016/j.apcata.2008.02.014 |
|
|
(c)
doi: 10.1016/j.jcat.2007.04.002 |
|
| [18] |
doi: 10.1016/j.tetlet.2005.06.169 |
| [19] |
(a)
doi: 10.3390/molecules25163595 |
|
(b)
doi: 10.1002/adsc.v345:5 |
|
| [20] |
(a)
doi: 10.1002/chem.v29.66 pmid: 21341727 |
|
(b)
doi: 10.1080/00397918908052713 pmid: 21341727 |
|
|
(c)
doi: 10.1021/ol902186d pmid: 21341727 |
|
|
(d)
doi: 10.1021/jo101865y pmid: 21341727 |
|
| [21] |
doi: 10.1080/17415993.2012.654392 |
| [22] |
(a)
doi: 10.1039/c2cc32013d |
|
(b)
doi: 10.1021/jo50016a019 |
|
| [23] |
doi: 10.1002/adsc.v357.10 |
| [24] |
(a)
doi: 10.1002/slct.v2.2 |
|
(b)
|
| [1] | 韦娜娜, 郭婉真, 鲁星, 任志强, 马豪杰, 张玉琦, 王记江, 韩波. 镁促进未保护的吲哚及喹喔啉衍生物的选择性还原[J]. 有机化学, 2026, 46(3): 1027-1038. |
| [2] | 许嘉龙, 孔泉, 陈建辉, 张明月, 姬小明, 程彪. 低价钨催化的有机反应研究进展[J]. 有机化学, 2026, 46(3): 759-772. |
| [3] | 张云一, 严含冰, 朱先进, 石永佳, 李俊鑫, 杨道山, 李旭锋. 镍催化烯烃与环硫鎓盐的还原串联芳烷基化反应[J]. 有机化学, 2026, 46(2): 653-663. |
| [4] | 曾依玲, 梁方鹏, 李辉, 刘荣荣, 李世清. α-氧代羧酸、炔和伯胺的多组分碳氢环化构筑异喹啉鎓骨架[J]. 有机化学, 2026, 46(2): 554-563. |
| [5] | 刘辉杨, 陈都, 苏毅进, 张鹏, 刘超. 频哪醇硼烷快速可控还原酰氯合成醛[J]. 有机化学, 2026, 46(2): 564-569. |
| [6] | 任才艺, 董文楠, 刘敬功, 杨爽, 房新强. 铑催化由环丙烯酮合成酰偶姻衍生物[J]. 有机化学, 2026, 46(2): 486-495. |
| [7] | 张天庆, 吴修明, 王丛丛. 低配位膦正离子化合物的研究进展[J]. 有机化学, 2025, 45(9): 3244-3254. |
| [8] | 许文, 罗美明, 曾小明. 铬催化三氟甲基烯烃的还原交叉偶联[J]. 有机化学, 2025, 45(9): 3401-3411. |
| [9] | 赵友学, 李兮若, 孟洛冰, 李春秀, 范贵生, 许建和. 醇脱氢酶/羰基还原酶与多底物分子适配性研究的进展★[J]. 有机化学, 2025, 45(9): 3175-3185. |
| [10] | 缪尹盛, 张磊, 陈建圻, 王丽丽, 段征. 双季鏻盐催化芳基炔烃与N-亚胺(异)喹啉叶立德合成吡唑并(异)喹啉衍生物[J]. 有机化学, 2025, 45(8): 2876-2884. |
| [11] | 杜顺利, 王雅雅, 郭家铭, 徐雪薇, 彭新华. 铜有机磷配合物催化芳烯烃β-C位的硼化反应研究[J]. 有机化学, 2025, 45(7): 2435-2443. |
| [12] | 胡春洋, Yasir Mumtaz, Khan Jahangir, 王雷秉, 蒋绿齐. HCF2SO2Na: 一种作为胺的硫代甲酰化试剂的新应用[J]. 有机化学, 2025, 45(6): 2171-2180. |
| [13] | 姚嫣, 付年凯. 光电化学金属催化研究进展[J]. 有机化学, 2025, 45(6): 1819-1837. |
| [14] | 田江艳, 李驰, 陈剑飞, 吕辉. 镍和钴催化卤代烃与醛酮/亚胺的还原偶联反应研究进展[J]. 有机化学, 2025, 45(6): 1946-1960. |
| [15] | 谢沈彤, 李文静, 刘钰, 陆熹, 师仁义. 镍催化多氟芳烃与烷基卤化物的还原烷基化反应[J]. 有机化学, 2025, 45(6): 2121-2127. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||