有机化学 ›› 2026, Vol. 46 ›› Issue (4): 1303-1319.DOI: 10.6023/cjoc202512048 上一篇 下一篇
综述与进展
收稿日期:2025-12-31
修回日期:2026-03-09
发布日期:2026-03-20
通讯作者:
焦雷
基金资助:Received:2025-12-31
Revised:2026-03-09
Published:2026-03-20
Contact:
Lei Jiao
Supported by:文章分享
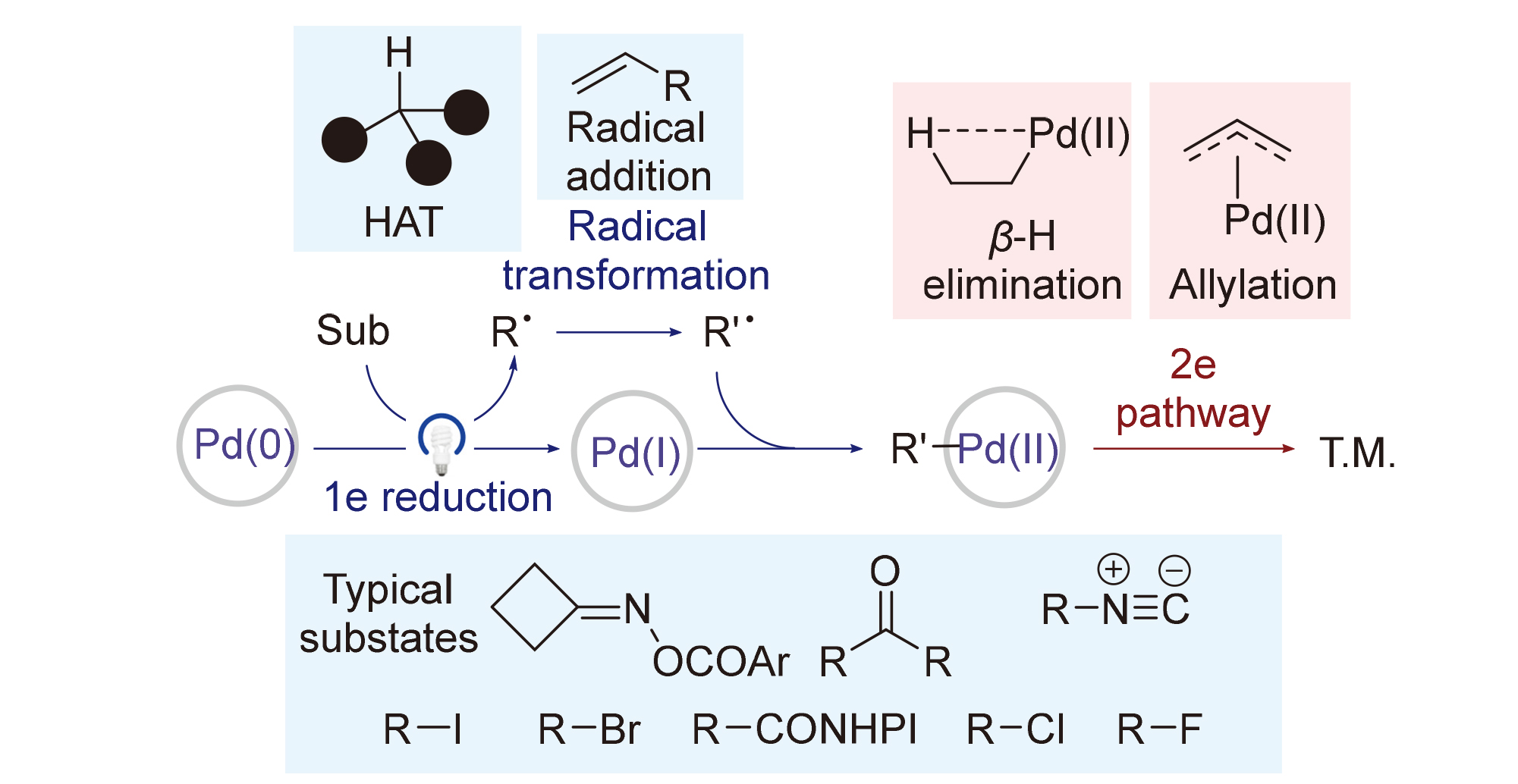
在现代有机合成的工具箱中, 钯催化的有机合成反应作为一类重要的反应类别, 长期以来被认为遵循经典双电子途径. 然而, 近二十年的研究, 特别是近期的突破表明, 在钯化学中存在着一个平行且同样重要的领域: 涉及单电子转移、卤原子转移和自由基机理的钯催化反应. 这一新反应范式不仅扩展了钯的催化能力, 使其能够活化传统上“惰性”的化学键, 也为在反应中实现新的化学选择性、区域选择性和立体选择性提供了可能. 本文旨在梳理这一前沿领域, 主要关注历经Pd(I)的自由基途径. 在这类反应中, Pd主要扮演自由基引发者的角色, 通过基态或激发态的Pd(0)发生氧化还原过程, 或者Pd(II)—C键的均裂产生自由基, 典型的Pd价态变化是Pd(0)-Pd(I)-Pd(II). 本文将从自由基产生方式的角度, 梳理包含Pd(I)中间体的自由基反应模式, 总结该领域近期研究进展, 并对相关反应机理进行评述.
谈逸凡, 焦雷. 经历Pd(I)途径的钯催化自由基反应: 反应模式简述和近期进展[J]. 有机化学, 2026, 46(4): 1303-1319.
Yi-Fan Tan, Lei Jiao. Palladium-Catalyzed Radical Reactions via Pd(I) Pathway: A Brief Summary of Reaction Patterns and Recent Progress[J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1303-1319.
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
Xie, B-Y.;
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [1] | 黄涎廷, 沈小倩, 王庆许, 柳忠全. 无催化剂及无溶剂光引发下氮杂芳烃的自由基脱羧酰基化反应[J]. 有机化学, 2026, 46(3): 951-960. |
| [2] | 谢赫男, 程瑶航, 安光辉. 位阻调控的吲哚C4位甲基化和C4甲基化/羰基重排反应[J]. 有机化学, 2026, 46(2): 475-485. |
| [3] | 赵倩倩, 魏培垚, 刘孙典, 张博鑫, 梁承远. 钯催化串联反应在含季碳中心的复杂天然产物全合成中的应用[J]. 有机化学, 2025, 45(9): 3289-3300. |
| [4] | 任天磊, 汪鑫, 丛欢. 蒽光二聚体衍生的手性单膦配体: 借助化学拆分的制备路线和不对称烯丙基胺化的合成应用[J]. 有机化学, 2025, 45(6): 2208-2221. |
| [5] | 姚团利, 王琳琳, 李涛. 钯催化多米诺Heck/分子间偶联反应合成3-烷基取代茚衍生物[J]. 有机化学, 2025, 45(4): 1342-1351. |
| [6] | 区洁晴, 屈培珍, 赵亮. 可见光介导下钯催化脂肪族α-溴代三氟甲基的脱溴还原反应[J]. 有机化学, 2025, 45(4): 1334-1341. |
| [7] | 龙涛, 何述钟, 李超. 自由基-极性交叉转化反应在天然产物全合成中的研究进展[J]. 有机化学, 2025, 45(3): 748-763. |
| [8] | 张朝威, 徐兵斌, 刘文龙, 赵敬, 段伟良. 钯催化不对称碳氢键活化合成平面手性二茂铁磺酰胺化合物[J]. 有机化学, 2025, 45(2): 707-716. |
| [9] | 梅明顺, 张扬会. 钯催化惰性亚甲基C(sp3)—H键分子间官能团化反应[J]. 有机化学, 2025, 45(2): 620-640. |
| [10] | 袁晨晖, 焦雷. 手性配体在钯催化配位辅助对映选择性C(sp3)—H键官能团化反应中的应用[J]. 有机化学, 2025, 45(2): 602-619. |
| [11] | 周艳, 王梦迪, 胡旭华, 郭文, 姚子健, 刘传祥, 郭勋祥. 钯催化苯胺、炔酯和炔烃合成多取代吡咯[J]. 有机化学, 2025, 45(12): 4481-4489. |
| [12] | 刘雯娟, 陈品红. 钯催化1,6-烯炔的环化反应研究[J]. 有机化学, 2024, 44(7): 2077-2091. |
| [13] | 张倩, 应垚璐, 张泓银, 徐林博, 林新奎, 黄晓雷. 钯催化SO2插入的炔丙基乙酸酯和碘代芳烃的还原偶联反应[J]. 有机化学, 2024, 44(6): 2033-2040. |
| [14] | 晏宇轩, 陆晚晴, 钱慧俊, 吕雷阳, 李志平. 钯催化偕二氟环丙烷开环与1,3-二羰基化合物的单/双氟烯丙基化反应[J]. 有机化学, 2024, 44(5): 1630-1640. |
| [15] | 罗东红, 李平, 陈志才, 杨佳怡, 孙梦凡, 陆居有. 钯催化邻-碳硼烷基吡啶卤化物交叉偶联合成邻-碳硼烷基联芳、氨基吡啶和炔基吡啶衍生物[J]. 有机化学, 2024, 44(5): 1568-1575. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
