有机化学 ›› 2026, Vol. 46 ›› Issue (3): 1039-1049.DOI: 10.6023/cjoc202508022 上一篇 下一篇
研究论文
马超a, 徐晓玲a, 骆翔a, 鲁瑞娟a, 虞国棋b,*( ), 蔡涛a,b,*(
), 蔡涛a,b,*( )
)
收稿日期:2025-08-25
修回日期:2025-10-15
发布日期:2025-11-27
基金资助:
Chao Maa, Xiaoling Xua, Xiang Luoa, Ruijuan Lua, Guoqi Yub,*( ), Tao Caia,b,*(
), Tao Caia,b,*( )
)
Received:2025-08-25
Revised:2025-10-15
Published:2025-11-27
Contact:
*E-mail: Supported by:文章分享
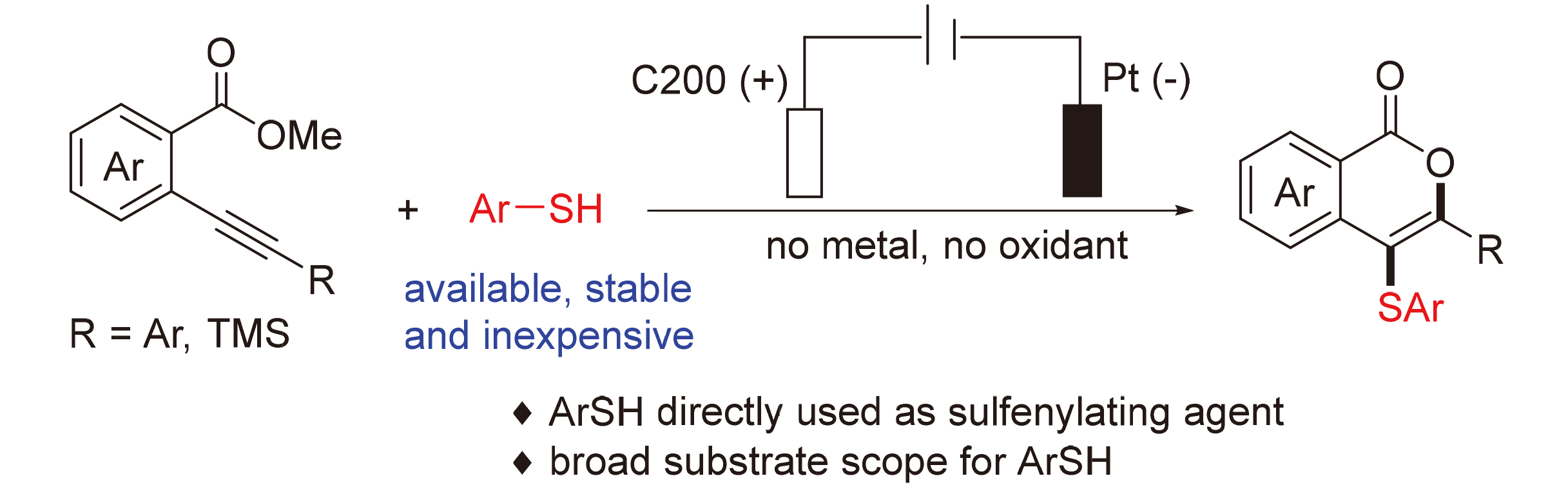
报道了一种在电化学条件下, 直接使用市售、稳定且廉价的硫酚作为硫化试剂构建4-硫代异香豆素类化合物的方法. 在优化后的反应条件下, 各种带吸电子基和给电子基的芳基硫酚均能与邻炔基苯甲酸酯顺利反应, 以中等到良好的收率得到相应的4-硫代异香豆素衍生物, 展现出优异的底物普适性. 初步机理研究表明, 该电化学促进的串联环化反应可能涉及自由基路径. 与文献方法相比, 该策略避免了使用对水敏感的预官能团化试剂, 如芳基硫氯(ArSCl)、二芳基二硫化物(ArSSAr)和N-硫代琥珀酰亚胺, 显著拓宽了底物适用范围.
马超, 徐晓玲, 骆翔, 鲁瑞娟, 虞国棋, 蔡涛. 电化学促进邻炔基苯甲酸酯与硫酚的自由基串联环化反应合成4-硫代异香豆素[J]. 有机化学, 2026, 46(3): 1039-1049.
Chao Ma, Xiaoling Xu, Xiang Luo, Ruijuan Lu, Guoqi Yu, Tao Cai. Electrochemical Synthesis of 4-Sulfenylisocoumarins via Radical Cascade Annulation of o-Alkynylbenzoates with Thiophenols[J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 1039-1049.
| Entry | Electrode (+/-) | Electrolyte | Solvent | Yieldb/% |
|---|---|---|---|---|
| 1 | Pt/Pt | Bu4NBF4 | CH3CN | Trace |
| 2 | GF/Pt | Bu4NBF4 | CH3CN | 24 |
| 3 | RVC/Pt | Bu4NBF4 | CH3CN | 12 |
| 4 | Carbon rod/Pt | Bu4NBF4 | CH3CN | 25 |
| 5 | C100/Pt | Bu4NBF4 | CH3CN | 45 |
| 6 | C200/Pt | Bu4NBF4 | CH3CN | 70 |
| 7 | C200/C100 | Bu4NBF4 | CH3CN | 46 |
| 8 | C100/C100 | Bu4NBF4 | CH3CN | 14 |
| 9 | C200/Pt | Bu4NBF4 | THF | Trace |
| 10 | C200/Pt | Bu4NBF4 | HFIP | 20 |
| 11 | C200/Pt | Bu4NBF4 | EtOH | 0 |
| 12 | C200/Pt | Bu4NBF4 | Acetone | 25 |
| 13 | C200/Pt | Bu4NBF4 | DMSO | 0 |
| 14 | C200/Pt | Bu4NBF4 | CH3CN/acetone (V∶V=10∶1) | 45 |
| 15 | C200/Pt | Bu4NBF4 | CH3CN/H2O (V∶V=10∶1) | 38 |
| 16 | C200/Pt | Bu4NBF4 | CH3CN/HFIP (V∶V=10∶1) | 82 |
| 17 | C200/Pt | Bu4NClO4 | CH3CN/HFIP (V∶V=10∶1) | 24 |
| 18 | C200/Pt | Bu4NPF6 | CH3CN/HFIP (V∶V=10∶1) | 15 |
| 19 | C200/Pt | Bu4NBr | CH3CN/HFIP (V∶V=10∶1) | Trace |
| 20 | C200/Pt | CF3SO3Li | CH3CN/HFIP (V∶V=10∶1) | 30 |
| 21 | C200/Pt | LiClO4 | CH3CN/HFIP (V∶V=10∶1) | 66 |
| 22c | C200/Pt | Bu4NBF4 | CH3CN/HFIP (V∶V=10∶1) | 70 |
| 23d | C200/Pt | Bu4NBF4 | CH3CN/HFIP (V∶V=10∶1) | 80 |
| 24e | C200/Pt | Bu4NBF4 | CH3CN/HFIP (V∶V=10∶1) | 48 |
| Entry | Electrode (+/-) | Electrolyte | Solvent | Yieldb/% |
|---|---|---|---|---|
| 1 | Pt/Pt | Bu4NBF4 | CH3CN | Trace |
| 2 | GF/Pt | Bu4NBF4 | CH3CN | 24 |
| 3 | RVC/Pt | Bu4NBF4 | CH3CN | 12 |
| 4 | Carbon rod/Pt | Bu4NBF4 | CH3CN | 25 |
| 5 | C100/Pt | Bu4NBF4 | CH3CN | 45 |
| 6 | C200/Pt | Bu4NBF4 | CH3CN | 70 |
| 7 | C200/C100 | Bu4NBF4 | CH3CN | 46 |
| 8 | C100/C100 | Bu4NBF4 | CH3CN | 14 |
| 9 | C200/Pt | Bu4NBF4 | THF | Trace |
| 10 | C200/Pt | Bu4NBF4 | HFIP | 20 |
| 11 | C200/Pt | Bu4NBF4 | EtOH | 0 |
| 12 | C200/Pt | Bu4NBF4 | Acetone | 25 |
| 13 | C200/Pt | Bu4NBF4 | DMSO | 0 |
| 14 | C200/Pt | Bu4NBF4 | CH3CN/acetone (V∶V=10∶1) | 45 |
| 15 | C200/Pt | Bu4NBF4 | CH3CN/H2O (V∶V=10∶1) | 38 |
| 16 | C200/Pt | Bu4NBF4 | CH3CN/HFIP (V∶V=10∶1) | 82 |
| 17 | C200/Pt | Bu4NClO4 | CH3CN/HFIP (V∶V=10∶1) | 24 |
| 18 | C200/Pt | Bu4NPF6 | CH3CN/HFIP (V∶V=10∶1) | 15 |
| 19 | C200/Pt | Bu4NBr | CH3CN/HFIP (V∶V=10∶1) | Trace |
| 20 | C200/Pt | CF3SO3Li | CH3CN/HFIP (V∶V=10∶1) | 30 |
| 21 | C200/Pt | LiClO4 | CH3CN/HFIP (V∶V=10∶1) | 66 |
| 22c | C200/Pt | Bu4NBF4 | CH3CN/HFIP (V∶V=10∶1) | 70 |
| 23d | C200/Pt | Bu4NBF4 | CH3CN/HFIP (V∶V=10∶1) | 80 |
| 24e | C200/Pt | Bu4NBF4 | CH3CN/HFIP (V∶V=10∶1) | 48 |
| [1] |
For selected reviews, see: (a) Saikia, P.; Gogoi, S. Adv. Synth. Catal. 2018, 360, 2063.
doi: 10.1002/adsc.v360.11 |
|
(b)
doi: 10.1016/j.tet.2023.133740 |
|
|
(c)
doi: 10.1002/asia.v18.5 |
|
|
|
|
|
(d)
doi: 10.1039/D3RA08149D |
|
|
(e)
doi: 10.1021/acs.jnatprod.4c01437 |
|
| [2] |
(a)
doi: 10.1016/j.bioorg.2021.104812 pmid: 20836556 |
|
(b)
doi: 10.1016/j.ejmech.2021.113793 pmid: 20836556 |
|
|
(c)
doi: 10.1021/jm100576z pmid: 20836556 |
|
| [3] |
(a)
doi: 10.1021/acs.joc.4c02696 |
|
(b)
doi: 10.1021/acs.orglett.2c00563 |
|
|
(c)
doi: 10.1021/acs.orglett.3c04082 |
|
|
(d)
doi: 10.1021/acs.joc.1c00271 |
|
|
(e)
doi: 10.1002/chem.v26.60 |
|
|
(f)
doi: 10.1039/D0RA06016J |
|
|
(g)
doi: 10.1039/D2NJ05567H |
|
| [4] |
(a)
doi: 10.1039/D4QO01367K pmid: 21721587 |
|
(b)
doi: 10.1002/adsc.v364.20 pmid: 21721587 |
|
|
(c)
doi: 10.1021/jo201211s pmid: 21721587 |
|
|
(d)
doi: 10.1021/acs.orglett.9b01046 pmid: 21721587 |
|
|
(e)
doi: 10.1016/j.tet.2011.12.003 pmid: 21721587 |
|
|
(f)
doi: 10.1002/ejoc.v2014.23 pmid: 21721587 |
|
| [5] |
doi: 10.1039/D4OB01137F |
| [6] |
For selected examples, see: (a)
doi: 10.1039/C8GC01337C |
|
(b)
doi: 10.1002/anie.v57.27 |
|
|
(c)
doi: 10.1002/adsc.v361.9 |
|
| [7] |
doi: 10.1002/celc.v5.23 |
| [8] |
For selected examples, see: (a) Wei, X.; Cai, T.; Zhang, Z.; Luo, Y.; Shang, T.; Shen, H.; Xu, H.; Yu, L.; Luo, X.; Yu, G.; Shen, R. J. Org. Chem. 2024, 89, 7804.
doi: 10.1021/acs.joc.4c00491 |
|
(b)
doi: 10.1039/D2OB01335E |
|
|
(c)
doi: 10.1002/adsc.v363.15 |
|
|
(d)
doi: 10.1021/acs.orglett.9b01510 |
|
| [9] |
doi: 10.1039/C9CC03996A |
| [10] |
doi: 10.1021/acs.orglett.8b03274 pmid: 30735046 |
| [11] |
doi: 10.1021/jo034308v |
| [1] | 盛馨瑶, 李鑫鑫, 邢潇, 潘玲, 刘群, 李亦菲. 基于2-硝基查尔酮与异氰基乙酸乙酯的串联环化反应合成α-咔啉酮类化合物[J]. 有机化学, 2025, 45(6): 2163-2170. |
| [2] | 杨之同, 宋恒谦, 雷盼, 闫嘉航, 谢卫青. Communesin生物碱核心五环骨架的高效合成[J]. 有机化学, 2025, 45(3): 1003-1008. |
| [3] | 何卫保, 易荣楠, 杨梓, 伍智林, 何卫民. Langlois试剂在电化学三氟甲基化反应中的应用进展[J]. 有机化学, 2025, 45(10): 3534-3545. |
| [4] | 李龙龙, 何欣悦, 周龙生, 曲亨通, 冯承涛, 徐坤. 硫氰酸铵促进的[3+3]环化反应合成5-芳基吡唑并[1,5-a]嘧啶[J]. 有机化学, 2024, 44(9): 2832-2840. |
| [5] | 邢运新, 闫登鸿, 温顺, 卜洁, 沈坤. 镍催化1,6-烯炔与芳基卤化物的反式还原芳基化环化[J]. 有机化学, 2024, 44(6): 1938-1948. |
| [6] | 杨帆, 方婷, 杨桂春, 高梦. 亚硝基苯参与的电化学串联环化反应构建喹啉/吡咯[J]. 有机化学, 2024, 44(3): 1021-1030. |
| [7] | 何蔺恒, 夏稳, 周玉祥, 于贤勇. 电催化N-芳基甘氨酸和苯并[e][1,2,3]噁噻嗪-2,2-二氧化物的串联脱羧环化反应[J]. 有机化学, 2024, 44(3): 997-1004. |
| [8] | 叶增辉, 刘华清, 张逢质. 有机光电催化合成研究进展[J]. 有机化学, 2024, 44(3): 840-870. |
| [9] | 陈远航, 何劲宇, 张博, 王延钊, 孔令轩, 钱伟烽, 王娜娜, 段闻喜, 欧阳妍妍, 朱翠菊, 徐浩. 不对称电化学有机合成[J]. 有机化学, 2024, 44(3): 748-779. |
| [10] | 樊思捷, 董武恒, 梁彩云, 王贵超, 袁瑶, 尹作栋, 张兆国. 可见光诱导的自由基环化反应构建4-芳基-1,2-二氢萘类化合物[J]. 有机化学, 2023, 43(9): 3277-3286. |
| [11] | 岁丹丹, 岑南楠, 龚若蕖, 陈阳, 陈文博. 无支持电解质条件下连续流电化学合成三氟甲基化氧化吲哚[J]. 有机化学, 2023, 43(9): 3239-3245. |
| [12] | 任志军, 罗维纬, 周俊. 银介导的N-芳基丙烯酰胺串联环化反应研究进展[J]. 有机化学, 2023, 43(6): 2026-2039. |
| [13] | 张俊颖, 赵晓静, 李干鹏, 何永辉. 室温下电化学合成保护型有机硼酸RB(dan)[J]. 有机化学, 2023, 43(5): 1815-1823. |
| [14] | 潘永周, 蒙秀金, 王迎春, 何慕雪. 电化学固定CO2构建羧酸衍生物的研究进展[J]. 有机化学, 2023, 43(4): 1416-1434. |
| [15] | 李靖鹏, 黄顺桃, 杨棋, 李伟强, 刘腾, 黄超. 利用连续流动技术合成(Z)-N-乙烯基取代N,O-缩醛[J]. 有机化学, 2023, 43(4): 1550-1558. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||