Chinese Journal of Organic Chemistry ›› 2023, Vol. 43 ›› Issue (8): 2926-2933.DOI: 10.6023/cjoc202303021 Previous Articles Next Articles
张素珍a,b, 张文文b, 杨慧b, 顾庆b,*( ), 游书力b,*(
), 游书力b,*( )
)
收稿日期:2023-03-15
修回日期:2023-03-28
发布日期:2023-04-07
基金资助:
Suzhen Zhanga,b, Wenwen Zhangb, Hui Yangb, Qing Gub( ), Shuli Youb(
), Shuli Youb( )
)
Received:2023-03-15
Revised:2023-03-28
Published:2023-04-07
Contact:
*E-mail: Supported by:Share
Suzhen Zhang, Wenwen Zhang, Hui Yang, Qing Gu, Shuli You. Rhodium-Catalyzed Enantioselective Spiroannulation of 2-Alkenylphenols with Alkynes[J]. Chinese Journal of Organic Chemistry, 2023, 43(8): 2926-2933.
| Entry | [Rh] | Solvent | Yieldb/% | eec/% |
|---|---|---|---|---|
| 1 | (R)-CpRh1 | CH3CN | 48 | 74 |
| 2 | (R)-CpRh2 | CH3CN | 60 | 40 |
| 3 | (R)-CpRh3 | CH3CN | 50 | 60 |
| 4 | (R)-CpRh4 | CH3CN | 39 | 56 |
| 5 | (R)-CpRh5 | CH3CN | 8 | 39 |
| 6 | (S)-SCpRh1 | CH3CN | 18 | 54 |
| 7 | (S)-SCpRh2 | CH3CN | 60 | 38 |
| 8d | (R)-CpRh1 | CH3CN | 52 | 76 |
| 9d | (R)-CpRh1 | tAmylOH | 60 | 75 |
| 10d | (R)-CpRh1 | 1,4-Dioxane | 14 | 83 |
| 11d | (R)-CpRh1 | THF | 21 | 80 |
| 12d | (R)-CpRh1 | DMF | 31 | 70 |
| 13d | (R)-CpRh1 | 1,4-Dioxane/tAmylOH | 42 | 77 |
| 14d | (R)-CpRh1 | THF/tAmylOH | 52 | 76 |
| 15d | (R)-CpRh1 | DMF/tAmylOH | 81 (77e) | 81 |
| Entry | [Rh] | Solvent | Yieldb/% | eec/% |
|---|---|---|---|---|
| 1 | (R)-CpRh1 | CH3CN | 48 | 74 |
| 2 | (R)-CpRh2 | CH3CN | 60 | 40 |
| 3 | (R)-CpRh3 | CH3CN | 50 | 60 |
| 4 | (R)-CpRh4 | CH3CN | 39 | 56 |
| 5 | (R)-CpRh5 | CH3CN | 8 | 39 |
| 6 | (S)-SCpRh1 | CH3CN | 18 | 54 |
| 7 | (S)-SCpRh2 | CH3CN | 60 | 38 |
| 8d | (R)-CpRh1 | CH3CN | 52 | 76 |
| 9d | (R)-CpRh1 | tAmylOH | 60 | 75 |
| 10d | (R)-CpRh1 | 1,4-Dioxane | 14 | 83 |
| 11d | (R)-CpRh1 | THF | 21 | 80 |
| 12d | (R)-CpRh1 | DMF | 31 | 70 |
| 13d | (R)-CpRh1 | 1,4-Dioxane/tAmylOH | 42 | 77 |
| 14d | (R)-CpRh1 | THF/tAmylOH | 52 | 76 |
| 15d | (R)-CpRh1 | DMF/tAmylOH | 81 (77e) | 81 |

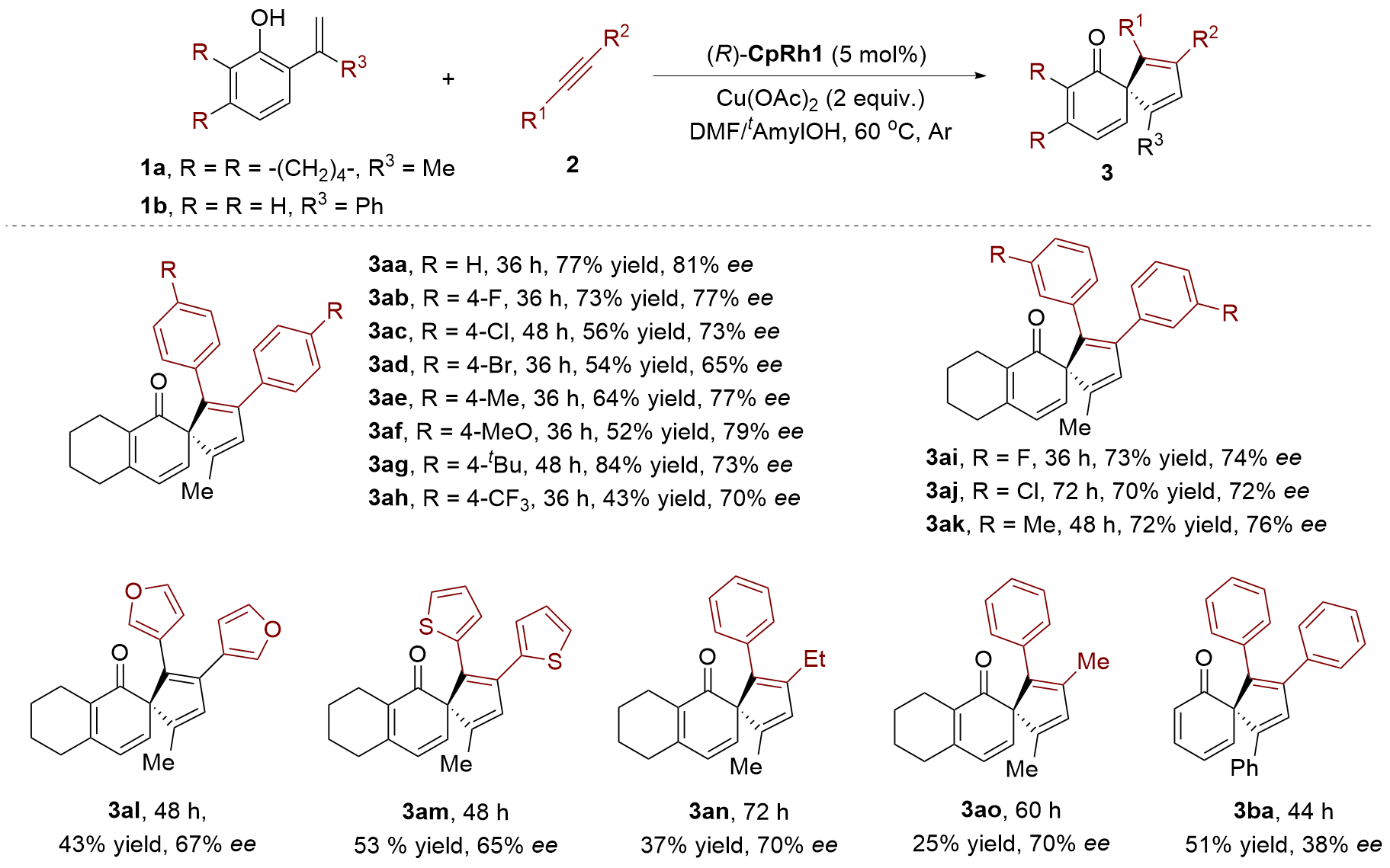
| [1] |
For reviews on transition-metal-catalyzed C—H activation/annul- ation reactions, see: (a) Satoh, T.; Miura, M. Chem.-Eur. J. 2010, 16, 11212.
doi: 10.1002/chem.v16:37 |
|
(b) Ackermann, L. Acc. Chem. Res. 2014, 47, 281.
doi: 10.1021/ar3002798 |
|
|
(c) Yang, Y.; Li, K.; Cheng, Y.; Wan, D.; Li, M.; You, J. Chem. Commun. 2016, 52, 2872.
doi: 10.1039/C5CC09180B |
|
|
(d) Duarah, G.; Kaishap, P. P.; Begum, T.; Gogoi, S. Adv. Synth. Catal. 2019, 361, 654.
doi: 10.1002/adsc.201800755 |
|
|
(d) Kuang, G.; Liu, G.; Zhang, X.; Lu, N.; Peng, Y.; Xiao, Q.; Zhou, Y. Synthesis 2020, 52, 993.
doi: 10.1055/s-0039-1690816 |
|
|
(e) Saha, A.; Shankar, M.; Sau, S.; Sahoo, A. K. Chem. Commun. 2022, 58, 4561.
doi: 10.1039/D2CC00172A |
|
|
Selected reviews on transition-metal-catalyzed C—H functionalizations, see: (f) Ji, Y.; Zhang, M.; Xing, M.; Cui, H.; Zhao, Q.; Zhang, C. Chin. J. Chem. 2021, 39, 391.
doi: 10.1002/cjoc.v39.2 |
|
|
(g) Liu, X.; Kuang, C.; Su, C. Acta Chim. Sinica 2022, 80, 1135. (in Chinese)
doi: 10.6023/A22040147 |
|
|
( 刘霞, 匡春香, 苏长会, 化学学报, 2022, 80, 1135.)
doi: 10.6023/A22040147 |
|
|
(h) Gu, H.; Jin, X.; Li, J.; Li, H.; Liu, J. Chin. J. Org. Chem. 2022, 42, 2682. (in Chinese)
doi: 10.6023/cjoc202204056 |
|
|
( 顾海春, 靳新新, 李嘉琪, 李贺, 刘景林, 有机化学, 2022, 42, 2682.)
doi: 10.6023/cjoc202204056 |
|
| [2] |
(a) Reddy Chidipudi, S.; Khan, I.; Lam, H. W. Angew. Chem., Int. Ed. 2012, 51, 12115.
doi: 10.1002/anie.201207170 |
|
(b) Dooley, J. D.; Reddy Chidipudi, S.; Lam, H. W. J. Am. Chem. Soc. 2013, 135, 10829.
doi: 10.1021/ja404867k |
|
|
(c) Reddy Chidipudi, S.; Burns, D. J.; Khan, I.; Lam, H. W. Angew. Chem., Int. Ed. 2015, 54, 13975.
doi: 10.1002/anie.v54.47 |
|
| [3] |
(a) Nan, J.; Zuo, Z.; Luo, L.; Bai, L.; Zheng, H.; Yuan, Y.; Liu, J.; Luan, X.; Wang, Y. J. Am. Chem. Soc. 2013, 135, 17306.
doi: 10.1021/ja410060e |
|
(b) Han, L.; Wang, H.; Luan, X. Org. Chem. Front. 2018, 5, 2453.
doi: 10.1039/C8QO00614H |
|
|
(c) Hao, J.; Ge, Y.; Yang, L.; Wang, J.; Luan, X. Tetrahedron Lett. 2021, 71, 153050.
doi: 10.1016/j.tetlet.2021.153050 |
|
|
(d) Li, K.; Bai, L.; Luan, X. Chin. J. Org. Chem. 2019, 39, 2211. (in Chinese)
doi: 10.6023/cjoc201903065 |
|
|
( 李锟雨, 白璐, 栾新军, 有机化学, 2019, 39, 2211.)
doi: 10.6023/cjoc201903065 |
|
|
(e) Li, H.; Cui, X. Chin. J. Org. Chem. 2020, 40, 543. (in Chinese)
doi: 10.6023/cjoc202000007 |
|
|
( 李欢, 崔秀灵, 有机化学, 2020, 40, 543.)
doi: 10.6023/cjoc202000007 |
|
| [4] |
(a) Zheng, J.; Wang, S.-B.; Zheng, C.; You, S.-L. J. Am. Chem. Soc. 2015, 137, 4880.
doi: 10.1021/jacs.5b01707 pmid: 25853771 |
|
(b) Zheng, C.; Zheng, J.; You, S.-L. ACS Catal. 2016, 6, 262.
doi: 10.1021/acscatal.5b01891 pmid: 25853771 |
|
| [5] |
Other examples of enantioselective C—H spiroannulation reactions with alkynes, see: (a) Zhang, M.; Huang, G. Chem. Eur. J. 2016, 22, 9356.
doi: 10.1002/chem.201600884 |
|
(b) Zheng, J.; Wang, S.-B.; Zheng, C.; You, S.-L. Angew. Chem., Int. Ed. 2017, 56, 4540.
doi: 10.1002/anie.201700021 |
|
|
(c) Duarah, G.; Kaishap, P. P.; Sarma, B.; Gogoi, S. Chem. Eur. J. 2018, 24, 10196.
doi: 10.1002/chem.v24.40 |
|
|
(d) Li, H.; Gontla, R.; Flegel, J.; Merten, C.; Ziegler, S.; Antonchick, A. P.; Waldmann, H. Angew. Chem., Int. Ed. 2019, 58, 307.
doi: 10.1002/anie.201811041 |
|
|
(e) Huang, Y.-Q.; Wu, Z.-J.; Zhu, L.; Gu, Q.; Lu, X.; You, S.-L.; Mei, T.-S. CCS Chem. 2022, 4, 3181.
doi: 10.31635/ccschem.021.202101376 |
|
|
(f) Wang, Y.; Huang, X.; Wang, Q.; Tang, Y.; Xu, S.; Li, Y. Org. Lett. 2021, 23, 757.
doi: 10.1021/acs.orglett.0c03982 |
|
|
(g) Wei, W.; Scheremetjew, A.; Ackermann, L. Chem. Sci. 2022, 13, 2783.
doi: 10.1039/D1SC07124F |
|
| [6] |
For C—H spiroannulation reactions of phenols with alkynes in a racemic manner, see: (a) Seoane, A.; Casanova, N.; Quinones, N.; Mascareñas, J. L.; Gulias, M. J. Am. Chem. Soc. 2014, 136, 7607.
doi: 10.1021/ja5034952 |
|
(b) Kujawa, S.; Best, D.; Burns, D. J.; Lam, H. W. Chem. Eur. J. 2014, 20, 8599.
doi: 10.1002/chem.201403454 |
|
|
(c) Zuo, Z.; Yang, X.; Liu, J.; Nan, J.; Bai, L.; Wang, Y.; Luan, X. J. Org. Chem. 2015, 80, 3349.
doi: 10.1021/acs.joc.5b00316 |
|
|
(d) Lin, P.-P.; Han, X.-L.; Ye, G.-H.; Li, J.-L.; Li, Q.; Wang, H. J. Org. Chem. 2019, 84, 12966.
doi: 10.1021/acs.joc.9b01750 |
|
|
(e) Liu, A.; Han, K.; Wu, X.-X.; Chen, S.; Wang, J. Chin. J. Chem. 2020, 38, 1257.
doi: 10.1002/cjoc.v38.11 |
|
|
(f) Liu, T.; Han, L.; Zhang, J.; Lu, G. J. Org. Chem. 2021, 86, 10484.
|
|
| [7] |
(a) Pettus, T. R. R.; Inoue, M.; Chen, X.-T.; Danishefsky, S. J. J. Am. Chem. Soc. 2000, 122, 6160.
doi: 10.1021/ja000521m pmid: 31008606 |
|
(b) Zhan, Z.-J.; Li, C.-P.; Shan, W.-G. J. Chem. Res. 2007, 1, 38.
pmid: 31008606 |
|
|
(c) Saito, Y.; Takashima, Y.; Kamada, A.; Suzuki, Y.; Suenaga, M.; Okamoto, Y.; Matsunaga, Y.; Hanai, R.; Kawahara, T.; Gong, X.; Tori, M.; Kuroda, C. Tetrahedron 2012, 68, 10011.
doi: 10.1016/j.tet.2012.09.042 pmid: 31008606 |
|
|
(d) Sandargo, B.; Michehl, M.; Praditya, D.; Steinmann, E.; Stadler, M.; Surup, F. Org. Lett. 2019, 21, 3286.
doi: 10.1021/acs.orglett.9b01017 pmid: 31008606 |
|
| [8] |
For selected reviews on Rh-catalyzed C—H functionalizations, see: (a) Ye, B.; Cramer, N. Acc. Chem. Res. 2015, 48, 1308.
doi: 10.1021/acs.accounts.5b00092 |
|
(b) Yoshino, T.; Satake, S.; Matsunaga, S. Chem. Eur. J. 2020, 26, 7346.
doi: 10.1002/chem.v26.33 |
|
|
(c) Shaaban, S.; Davies, C.; Waldmann, H. Eur. J. Org. Chem. 2020, 6512.
|
|
|
(d) Mas-Rosello, J.; Herraiz, A. G.; Audic, B.; Laverny, A.; Cramer, N. Angew. Chem., Int. Ed. 2021, 60, 13198.
doi: 10.1002/anie.v60.24 |
|
|
(e) Davies, C.; Shaaban, S.; Waldmann, H. Trends Chem. 2022, 4, 318.
doi: 10.1016/j.trechm.2022.01.005 |
|
| [9] |
(a) Ye, B.; Cramer, N. Science 2012, 338, 504.
doi: 10.1126/science.1226938 pmid: 27070297 |
|
(b) Ye, B.; Cramer, N. J. Am. Chem. Soc. 2013, 135, 636.
doi: 10.1021/ja311956k pmid: 27070297 |
|
|
(c) Zheng, J.; Cui, W.-J.; Zheng, C.; You, S.-L. J. Am. Chem. Soc. 2016, 138, 5242.
doi: 10.1021/jacs.6b02302 pmid: 27070297 |
| [1] | Wenwen Chen, Qin Zhang, Songyue Zhang, Fangfang Huang, Xinyin Zhang, Jianfeng Jia. Visible Light Promoted Coupling Reaction of Alkynyl Iodide and Sodium Sulphinate without Photocatalyst [J]. Chinese Journal of Organic Chemistry, 2024, 44(2): 584-592. |
| [2] | Wenfeng Bei, Jian Pan, Dongmei Ran, Yilin Liu, Zhen Yang, Ruokun Feng. Cobalt-Catalyzed [4+2] Annulation of Indole Carboxamide with Diynes and Monoacetylene: Direct Access to γ-Carbolinones [J]. Chinese Journal of Organic Chemistry, 2023, 43(9): 3226-3238. |
| [3] | Changjun Liu, Huiling Hu, Chenghong Liu, Chaojie Zhu, Tiandi Tang. Pd Supported on Mesoporous ETS-10 Zeolite Catalyst with Superior Catalytic Performances in Synthesizing 1,2-Diones from the Oxidation of Internal Alkynes [J]. Chinese Journal of Organic Chemistry, 2023, 43(8): 2953-2960. |
| [4] | Jiamin Ma, Jiaoxiong Li, Qiansen Meng, Xianghua Zeng. Advances on the Radical Sulfonation of Alkynes [J]. Chinese Journal of Organic Chemistry, 2023, 43(6): 2040-2052. |
| [5] | Kai Lu, Haoqi Qu, Xi Chen, Hui Qiu, Jing Zheng, Mengtao Ma. Catalyst-Free and Solvent-Free Hydroboration of Alkynes and Alkenes with Catecholborane [J]. Chinese Journal of Organic Chemistry, 2023, 43(6): 2197-2205. |
| [6] | Sida Li, Xing-Zhong Shu, Lipeng Wu. Zirconium and Titanium Mediated Hydroboration of Alkenes and Alkynes [J]. Chinese Journal of Organic Chemistry, 2023, 43(5): 1751-1760. |
| [7] | Zhihao Chen, Qi Fan, Biaolin Yin, Qingjiang Li, Honggen Wang. Progress in the Syntheses of α-Boryl Carbonyl Compounds [J]. Chinese Journal of Organic Chemistry, 2023, 43(5): 1706-1712. |
| [8] | Shiquan Gao, Chuangjun Liu, Junfeng Yang, Junliang Zhang. Cobalt-Catalyzed Electrochemical Reductive Coupling of Alkynes and Alkenes [J]. Chinese Journal of Organic Chemistry, 2023, 43(4): 1559-1565. |
| [9] | Peng Liu, Fuming Zhong, Lihao Liao, Weiqiang Tan, Xiaodan Zhao. Progress in the Construction of Spirocyclohexadienones via Alkyne-Involving Dearomatization [J]. Chinese Journal of Organic Chemistry, 2023, 43(12): 4019-4035. |
| [10] | Junxiu Liang, Yazhou Liu, Amu Wang, Yanchao Wu, Xiaofeng Ma, Huijing Li. Dearomatization of Halonaphthols via an Intermolecular [4+1] Spiroannulation with in situ Formed Aza-ortho-quinone Methides [J]. Chinese Journal of Organic Chemistry, 2023, 43(11): 3888-3899. |
| [11] | Chong Tian, Qi Sun, Junfeng Wang, Qiao Chen, Zhiguo Wen, Maxim Borzov, Wanli Nie. E-Stereospecific 1,1-Carboboration of Terminal Arylalkynes with [IB(C6F5)3]– [J]. Chinese Journal of Organic Chemistry, 2023, 43(1): 338-344. |
| [12] | Qi Sun, Zeying Sun, Ze Yu, Guangwei Wang. Nickel-Catalyzed Stereoselective Aryl-Difluoroalkylation of Alkynes [J]. Chinese Journal of Organic Chemistry, 2022, 42(8): 2515-2520. |
| [13] | Fei Chen, Sheng Tao, Ning Liu, Bin Dai. CNN-Type Binuclear Cu(I) Complexes Catalyzed Direct Carboxylation via the Fixation of CO2 at Room Temperature [J]. Chinese Journal of Organic Chemistry, 2022, 42(8): 2471-2480. |
| [14] | Huili Liu, Chaojie Zhu, Tiandi Tang. Acidic Zeolite HBeta Catalyzed Friedel-Crafts Alkenylation Reaction [J]. Chinese Journal of Organic Chemistry, 2022, 42(6): 1792-1798. |
| [15] | Liwen Ma, Xiaoye Wei, Zilin Zhao, Ang Zhao, Xiangwen Deng, Bingnan Huo, Gang Ma, Chunfang Zhang. Theoretical Study on the Catalytic Mechanism of Copper with Various Valence for the Terminal Alkyne Coupling Reaction [J]. Chinese Journal of Organic Chemistry, 2022, 42(6): 1811-1819. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||