化学学报 ›› 2011, Vol. 69 ›› Issue (21): 2627-2631.DOI: 10.6023/A1007192B 上一篇 下一篇
研究论文
夏笑虹,石磊,何月德,杨丽,刘洪波*
XIA Xiao-Hong, SHI Lei, HE Yue-De, YANG Li, LIU Hong-Bo
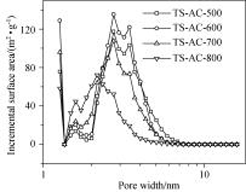
以烟杆为原料, 氢氧化钾为活化剂, 通过调节炭化温度(500~800 ℃温度范围)在相同活化条件下制备了具有不同孔隙结构的活性炭材料. N2吸附测试表明随着炭化温度降低, 活性炭的比表面积和总孔容先增大后减小, 中孔比表面积和平均孔径却一直增大. 其中600 ℃炭化样品经KOH活化后可制得比表面积为3333 m2•g-1, 总孔容为2.47 cm3• g-1, 中孔孔容达2.11 cm3•g-1的高中孔率高比表面积活性炭材料. 采用直流充放电法、交流阻抗法和循环伏安法测定上述多孔炭为电极材料的双电层电容器的电化学性能, 结果表明: 炭化温度不同的烟杆基活性炭电极均表现出良好的功率特性, 充放电流增大50倍, 容量保持率均在80%左右, 其中TS-AC-600活性炭电极在有机电解液中1 mA•cm-2充放电时, 比电容达到190 F•g-1. 较高的中孔率和较大的平均孔径使得烟杆基活性炭电极具有良好的高倍率充放电性能.