Chinese Journal of Organic Chemistry ›› 2021, Vol. 41 ›› Issue (10): 3844-3879.DOI: 10.6023/cjoc202106001 Previous Articles Next Articles
Special Issue: 南开大学化学学科创立100周年; 热点论文虚拟合集
REVIEWS
收稿日期:2021-06-01
修回日期:2021-07-22
发布日期:2021-08-24
通讯作者:
刘天飞
基金资助:
Zijie Zhoua, Xiangmei Konga, Tianfei Liua,b( )
)
Received:2021-06-01
Revised:2021-07-22
Published:2021-08-24
Contact:
Tianfei Liu
Supported by:Share
Zijie Zhou, Xiangmei Kong, Tianfei Liu. Applications of Proton-Coupled Electron Transfer in Organic Synthesis[J]. Chinese Journal of Organic Chemistry, 2021, 41(10): 3844-3879.
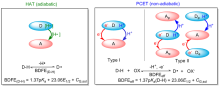
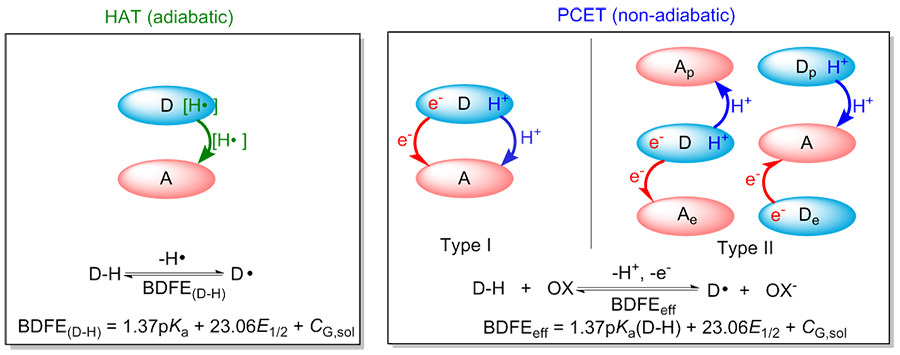
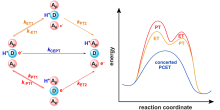
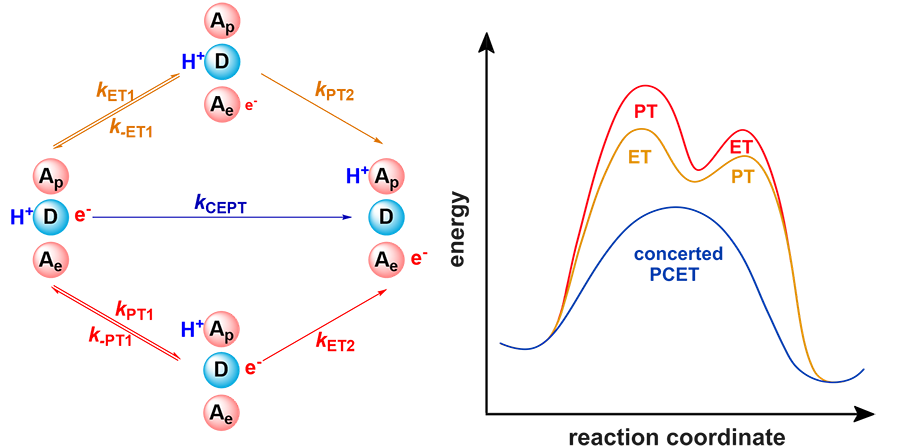

| [1] |
Hammes-Schiffer, S.; Iordanova, N. Biochim. Biophys. Acta 2004, 1655, 29.
pmid: 15100013 |
| [2] |
Hammes-Schiffer, S. Acc. Chem. Res. 2009, 42, 1881.
doi: 10.1021/ar9001284 |
| [3] |
Mayer, J. M. Annu. Rev. Phys. Chem. 2004, 55, 363.
doi: 10.1146/annurev.physchem.55.091602.094446 |
| [4] |
Mayer, J. M.; Rhile, I. J. Biochim. Biophys. Acta 2004, 1655, 51.
|
| [5] |
Rhile, I. J.; Markle, T. F.; Nagao, H.; DiPasquale, A. G.; Lam, O. P.; Lockwood, M. A.; Rotter, K.; Mayer, J. M. J. Am. Chem. Soc. 2006, 128, 6075.
pmid: 16669677 |
| [6] |
Tyburski, R.; Liu, T.; Glover, S. D.; Hammarström, L. J. Am. Chem. Soc. 2021, 143, 560.
doi: 10.1021/jacs.0c09106 pmid: 33405896 |
| [7] |
Gentry, E. C.; Knowles, R. R. Acc. Chem. Res. 2016, 49, 1546.
doi: 10.1021/acs.accounts.6b00272 |
| [8] |
Yang, J.-D.; Ji, P.; Xue, X.-S.; Cheng, J.-P. J. Am. Chem. Soc. 2018, 140, 8611.
doi: 10.1021/jacs.8b04104 |
| [9] |
Wu, X.; Zhu, C. Chin. J. Chem. 2019, 37, 171.
|
| [10] |
Symes, M. D.; Surendranath, Y.; Lutterman, D. A.; Nocera, D. G. J. Am. Chem. Soc. 2011, 133, 5174.
doi: 10.1021/ja110908v pmid: 21413703 |
| [11] |
Guo, Y.; Liu, Y.; Qi, J.; Li, H.; He, L.; Lu, L.; Liu, C.; Gong, L.; Zhao, D.; Yang, Z. Acta Chim. Sinica 2017, 75, 914. (in Chinese)
doi: 10.6023/A17050214 |
|
(郭宇, 刘瑜, 戚娟娟, 李慧, 赫兰兰, 卢丽男, 刘翠, 宫利东, 赵东霞, 杨忠志, 化学学报, 2017, 75, 914.)
doi: 10.6023/A17050214 |
|
| [12] |
Odella, E.; Mora, S. J.; Wadsworth, B. L.; Huynh, M. T.; Goings, J. J.; Liddell, P. A.; Groy, T. L.; Gervaldo, M.; Sereno, L. E.; Gust, D.; Moore, T. A.; Moore, G. F.; Hammes-Schiffer, S.; Moore, A. L. J. Am. Chem. Soc. 2018, 140, 15450.
doi: 10.1021/jacs.8b09724 |
| [13] |
Liu, T.; Guo, M.; Orthaber, A.; Lomoth, R.; Lundberg, M.; Ott, S.; Hammarström, L. Nat. Chem. 2018, 10, 881.
doi: 10.1038/s41557-018-0076-x |
| [14] |
Liu, T.; Tyburski, R.; Wang, S.; Fernandez-Teran, R.; Ott, S.; Hammarström, L. J. Am. Chem. Soc. 2019, 141, 17245.
doi: 10.1021/jacs.9b08189 |
| [15] |
Bourrez, M.; Steinmetz, R.; Ott, S.; Gloaguen, F.; Hammarström, L. Nat. Chem. 2015, 7, 140.
doi: 10.1038/nchem.2157 |
| [16] |
Jackson, M. N.; Pegis, M. L.; Surendranath, Y. ACS Cent. Sci. 2019, 5, 831.
doi: 10.1021/acscentsci.9b00114 |
| [17] |
Wenger, O. S. Chem.-Eur. J. 2011, 17, 11692.
doi: 10.1002/chem.v17.42 |
| [18] |
Wenger, O. S. Acc. Chem. Res. 2013, 46, 1517.
doi: 10.1021/ar300289x |
| [19] |
Chen, Z.; Wang, T.; Sun, T.; Chen, Z.; Sheng, T.; Hong, Y.-H.; Nan, Z.-A.; Zhu, J.; Zhou, Z.-Y.; Xia, H.; Sun, S.-G. Chin. J. Chem. 2018, 36, 1161.
doi: 10.1002/cjoc.v36.12 |
| [20] |
Binstead, R. A.; Moyer, B. A.; Samuels, G. J.; Meyer, T. J. J. Am. Chem. Soc. 1981, 103, 2897.
doi: 10.1021/ja00400a083 |
| [21] |
Liu, F.; Concepcion, J. J.; Jurss, J. W.; Cardolaccia, T.; Templeton, J. L.; Meyer, T. J. Inorg. Chem. 2008, 47, 1727.
doi: 10.1021/ic701249s |
| [22] |
Bonin, J.; Robert, M. Photochem. Photobiol. 2011, 87, 1190.
doi: 10.1111/php.2011.87.issue-6 |
| [23] |
Hoffmann, N. Eur. J. Org. Chem. 2017, 2017, 1982.
doi: 10.1002/ejoc.201601445 |
| [24] |
Hammes-Schiffer, S.; Soudackov, A. V. J. Phys. Chem. B 2008, 112, 14108.
doi: 10.1021/jp805876e |
| [25] |
Sanderson, R. T. Polar Covalence, Academic Press, New York, 1983, p. 46.
|
| [26] |
Sanderson, R. T. Chemical Bonds and Bond Energy, Academic Press, New York, 1976, p. 185.
|
| [27] |
Marcus, R. A.; Sutin, N. Biochim. Biophys. Acta 1985, 811, 265.
|
| [28] |
Bolton, J. R.; Archer, M. D. In Electron Transfer in Inorganic, Organic, and Biological Systems, American Chemical Society, Washington, 1991, pp. 7-23.
|
| [29] |
Marcus, R. A. J. Chem. Phys. 1956, 24, 966.
doi: 10.1063/1.1742723 |
| [30] |
Borgis, D.; Hynes, J. T. J. Phy. Chem. 1996, 100, 1118.
doi: 10.1021/jp9522324 |
| [31] |
Krishtalik, L. I. Biochim. Biophys. Acta 2000, 1458, 6.
pmid: 10812022 |
| [32] |
Soudackov, A.; Hammes-Schiffer, S. J. Chem. Phys. 1999, 111, 4672.
doi: 10.1063/1.479229 |
| [33] |
Soudackov, A.; Hammes-Schiffer, S. J. Chem. Phys. 2000, 113, 2385.
doi: 10.1063/1.482053 |
| [34] |
Hammes-Schiffer, S.; Stuchebrukhov, A. A. Chem. Rev. 2010, 110, 6939.
doi: 10.1021/cr1001436 pmid: 21049940 |
| [35] |
Huynh, M. H. V.; Meyer, T. J. Chem. Rev. 2007, 107, 5004.
pmid: 17999556 |
| [36] |
Warren, J. J.; Tronic, T. A.; Mayer, J. M. Chem. Rev. 2010, 110, 6961.
doi: 10.1021/cr100085k |
| [37] |
Weinberg, D. R.; Gagliardi, C. J.; Hull, J. F.; Murphy, C. F.; Kent, C. A.; Westlake, B. C.; Paul, A.; Ess, D. H.; McCafferty, D. G.; Meyer, T. J. Chem. Rev. 2012, 112, 4016.
doi: 10.1021/cr200177j pmid: 22702235 |
| [38] |
Waidmann, C. R.; Miller, A. J. M.; Ng, C.-W. A.; Scheuermann, M. L.; Porter, T. R.; Tronic, T. A.; Mayer, J. M. Energy Environ. Sci. 2012, 5, 7771.
doi: 10.1039/c2ee03300c |
| [39] |
Darcy, J. W.; Koronkiewicz, B.; Parada, G. A.; Mayer, J. M. Acc. Chem. Res. 2018, 51, 2391.
doi: 10.1021/acs.accounts.8b00319 |
| [40] |
Morton, C. M.; Zhu, Q.; Ripberger, H.; Troian-Gautier, L.; Toa, Z. S. D.; Knowles, R. R.; Alexanian, E. J. J. Am. Chem. Soc. 2019, 141, 13253.
doi: 10.1021/jacs.9b06834 pmid: 31356059 |
| [41] |
Tarantino, K. T.; Liu, P.; Knowles, R. R. J. Am. Chem. Soc. 2013, 135, 10022.
doi: 10.1021/ja404342j pmid: 23796403 |
| [42] |
Skone, J. H.; Soudackov, A. V.; Hammes-Schiffer, S. J. Am. Chem. Soc. 2006, 128, 16655.
doi: 10.1021/ja0656548 |
| [43] |
Liu, T.; Tyburski, R.; Wang, S.; Fernández-Terán, R.; Ott, S.; Hammarström, L. J. Am. Chem. Soc. 2019, 141, 17245.
doi: 10.1021/jacs.9b08189 |
| [44] |
Huang, T.; Rountree, E. S.; Traywick, A. P.; Bayoumi, M.; Dempsey, J. L. J. Am. Chem. Soc. 2018, 140, 14655.
doi: 10.1021/jacs.8b07102 pmid: 30362720 |
| [45] |
Darcy, J. W.; Kolmar, S. S.; Mayer, J. M. J. Am. Chem. Soc. 2019, 141, 10777.
doi: 10.1021/jacs.9b04303 |
| [46] |
Zhang, J.; Yang, J.-D.; Cheng, J.-P. Chem. Sci. 2020, 11, 3672.
doi: 10.1039/C9SC05883D |
| [47] |
Chen, X.; Engle, K. M.; Wang, D. H.; Yu, J. Q. Angew. Chem., Int. Ed. 2009, 48, 5094.
doi: 10.1002/anie.v48:28 |
| [48] |
Giri, R.; Shi, B. F.; Engle, K. M.; Maugel, N.; Yu, J. Q. Chem. Soc. Rev. 2009, 38, 3242.
doi: 10.1039/b816707a |
| [49] |
Engle, K. M.; Mei, T.-S.; Wasa, M.; Yu, J.-Q. Acc. Chem. Res. 2012, 45, 788.
doi: 10.1021/ar200185g |
| [50] |
Sun, C. L.; Li, B. J.; Shi, Z. J. Chem. Rev. 2011, 111, 1293.
doi: 10.1021/cr100198w |
| [51] |
Markle, T. F.; Darcy, J. W.; Mayer, J. M. Sci. Adv. 2018, 4, eaat5776.
doi: 10.1126/sciadv.aat5776 |
| [52] |
Sayfutyarova, E. R.; Goldsmith, Z. K.; Hammes-Schiffer, S. J. Am. Chem. Soc. 2018, 140, 15641.
doi: 10.1021/jacs.8b10461 pmid: 30383371 |
| [53] |
Sayfutyarova, E. R.; Lam, Y. C.; Hammes-Schiffer, S. J. Am. Chem. Soc. 2019, 141, 15183.
doi: 10.1021/jacs.9b06849 pmid: 31464122 |
| [54] |
Uraguchi, D.; Torii, M.; Ooi, T. ACS Catal. 2017, 7, 2765.
doi: 10.1021/acscatal.7b00265 |
| [55] |
Shevchenko, G. A.; Oppelaar, B.; List, B. Angew. Chem., Int. Ed. 2018, 57, 10756.
doi: 10.1002/anie.v57.33 |
| [56] |
Leng, L.; Ready, J. M. ACS Catal. 2020, 10, 13196.
doi: 10.1021/acscatal.0c04519 |
| [57] |
Wu, Z. J.; Xu, H. C. Angew. Chem., Int. Ed. 2017, 56, 4734.
doi: 10.1002/anie.201701329 |
| [58] |
Lennox, A. J. J.; Goes, S. L.; Webster, M. P.; Koolman, H. F.; Djuric, S. W.; Stahl, S. S. J. Am. Chem. Soc. 2018, 140, 11227.
doi: 10.1021/jacs.8b08145 |
| [59] |
Zhang, Z.; Zhang, L.; Cao, Y.; Li, F.; Bai, G.; Liu, G.; Yang, Y.; Mo, F. Org. Lett. 2019, 21, 762.
doi: 10.1021/acs.orglett.8b04010 |
| [60] |
Sharma, S.; Roy, A.; Shaw, K.; Bisai, A.; Paul, A. J. Org. Chem. 2020, 85, 14926.
doi: 10.1021/acs.joc.0c01621 |
| [61] |
He, M. X.; Mo, Z. Y.; Wang, Z. Q.; Cheng, S. Y.; Xie, R. R.; Tang, H. T.; Pan, Y. M. Org. Lett. 2020, 22, 724.
doi: 10.1021/acs.orglett.9b04549 |
| [62] |
Tanwar, L.; Borgel, J.; Ritter, T. J. Am. Chem. Soc. 2019, 141, 17983.
doi: 10.1021/jacs.9b09496 |
| [63] |
Wang, Z.; Liu, Q.; Ji, X.; Deng, G.-J.; Huang, H. ACS Catal. 2019, 10, 154.
doi: 10.1021/acscatal.9b04411 |
| [64] |
Wu, Y.; Chen, J.-Y.; Ning, J.; Jiang, X.; Deng, J.; Deng, Y.; Xu, R.; He, W.-M. Green Chem. 2021, 23, 3950.
doi: 10.1039/D1GC00562F |
| [65] |
Peng, S.; Lin, Y.; He, W.-M. Chin. J. Org. Chem. 2020, 40, 541. (in Chinese)
doi: 10.6023/cjoc202000006 |
|
(彭莎, 林英武, 何卫民, 有机化学, 2020, 40, 541.)
doi: 10.6023/cjoc202000006 |
|
| [66] |
Luo, Y. R. Handbook of Bond Dissociation Energies in Organic Compounds, CRC Press, Boca Raton, 2003.
|
| [67] |
Choi, G. J.; Knowles, R. R. J. Am. Chem. Soc. 2015, 137, 9226.
doi: 10.1021/jacs.5b05377 |
| [68] |
Miller, D. C.; Choi, G. J.; Orbe, H. S.; Knowles, R. R. J. Am. Chem. Soc. 2015, 137, 13492.
doi: 10.1021/jacs.5b09671 pmid: 26439818 |
| [69] |
Tarantino, K. T.; Miller, D. C.; Callon, T. A.; Knowles, R. R. J. Am. Chem. Soc. 2015, 137, 6440.
doi: 10.1021/jacs.5b03428 pmid: 25945955 |
| [70] |
Choi, G. J.; Zhu, Q.; Miller, D. C.; Gu, C. J.; Knowles, R. R. Nature 2016, 539, 268.
doi: 10.1038/nature19811 |
| [71] |
Gentry, E. C.; Rono, L. J.; Hale, M. E.; Matsuura, R.; Knowles, R. R. J. Am. Chem. Soc. 2018, 140, 3394.
doi: 10.1021/jacs.7b13616 pmid: 29432006 |
| [72] |
Zhu, Q.; Graff, D. E.; Knowles, R. R. J. Am. Chem. Soc. 2018, 140, 741.
doi: 10.1021/jacs.7b11144 |
| [73] |
Nguyen, S. T.; Zhu, Q.; Knowles, R. R. ACS Catal. 2019, 9, 4502.
doi: 10.1021/acscatal.9b00966 pmid: 32292642 |
| [74] |
Roos, C. B.; Demaerel, J.; Graff, D. E.; Knowles, R. R. J. Am. Chem. Soc. 2020, 142, 5974.
doi: 10.1021/jacs.0c01332 |
| [75] |
Zhou, Z.; Li, Y.; Han, B.; Gong, L.; Meggers, E. Chem. Sci. 2017, 8, 5757.
doi: 10.1039/C7SC02031G |
| [76] |
Yuan, W.; Zhou, Z.; Gong, L.; Meggers, E. Chem. Commun. 2017, 53, 8964.
doi: 10.1039/C7CC04941B |
| [77] |
Jia, J.; Ho, Y. A.; Bulow, R. F.; Rueping, M. Chem.-Eur. J. 2018, 24, 14054.
doi: 10.1002/chem.v24.53 |
| [78] |
Moon, Y.; Jang, E.; Choi, S.; Hong, S. Org. Lett. 2018, 20, 240.
doi: 10.1021/acs.orglett.7b03600 |
| [79] |
Zheng, S.; Gutierrez-Bonet, A.; Molander, G. A. Chem 2019, 5, 339.
doi: 10.1016/j.chempr.2018.11.014 |
| [80] |
Zhou, C.; Lei, T.; Wei, X. Z.; Ye, C.; Liu, Z.; Chen, B.; Tung, C. H.; Wu, L. Z. J. Am. Chem. Soc. 2020, 142, 16805.
doi: 10.1021/jacs.0c07600 |
| [81] |
Zhu, L.; Xiong, P.; Mao, Z. Y.; Wang, Y. H.; Yan, X.; Lu, X.; Xu, H. C. Angew. Chem., Int. Ed. 2016, 55, 2226.
doi: 10.1002/anie.201510418 |
| [82] |
Xiong, P.; Xu, H. H.; Xu, H. C. J. Am. Chem. Soc. 2017, 139, 2956.
doi: 10.1021/jacs.7b01016 pmid: 28199102 |
| [83] |
Hu, X.; Zhang, G.; Bu, F.; Nie, L.; Lei, A. ACS Catal. 2018, 8, 9370.
doi: 10.1021/acscatal.8b02847 |
| [84] |
Wang, F.; Gerken, J. B.; Bates, D. M.; Kim, Y. J.; Stahl, S. S. J. Am. Chem. Soc. 2020, 142, 12349.
doi: 10.1021/jacs.0c04626 pmid: 32520537 |
| [85] |
Xu, Z.; Huang, Z.; Li, Y.; Kuniyil, R.; Zhang, C.; Ackermann, L.; Ruan, Z. Green Chem. 2020, 22, 1099.
doi: 10.1039/C9GC03901E |
| [86] |
Taylor, R. J. K.; Reid, M.; Foot, J.; Raw, S. A. Acc. Chem. Res. 2005, 38, 851.
doi: 10.1021/ar050113t |
| [87] |
Uyanik, M.; Ishihara, K. Chem. Commun. 2009, 2086.
|
| [88] |
Ciriminna, R.; Pagliaro, M. Org. Process Res. Dev. 2010, 14, 245-251.
doi: 10.1021/op900059x |
| [89] |
Hoover, J. M.; Stahl, S. S. J. Am. Chem. Soc. 2011, 133, 16901.
doi: 10.1021/ja206230h |
| [90] |
Greene, J. F.; Hoover, J. M.; Mannel, D. S.; Root, T. W.; Stahl, S. S. Org. Process Res. Dev. 2013, 17, 1247.
doi: 10.1021/op400207f |
| [91] |
Rahimi, A.; Azarpira, A.; Kim, H.; Ralph, J.; Stahl, S. S. J. Am. Chem. Soc. 2013, 135, 6415.
doi: 10.1021/ja401793n |
| [92] |
Steves, J. E.; Preger, Y.; Martinelli, J. R.; Welch, C. J.; Root, T. W.; Hawkins, J. M.; Stahl, S. S. Org. Process Res. Dev. 2015, 19, 1548.
doi: 10.1021/acs.oprd.5b00179 |
| [93] |
Rafiee, M.; Konz, Z. M.; Graaf, M. D.; Koolman, H. F.; Stahl, S. S. ACS Catal. 2018, 8, 6738.
doi: 10.1021/acscatal.8b01640 |
| [94] |
Rafiee, M.; Alherech, M.; Karlen, S. D.; Stahl, S. S. J. Am. Chem. Soc. 2019, 141, 15266.
doi: 10.1021/jacs.9b07243 |
| [95] |
Ota, E.; Wang, H.; Frye, N. L.; Knowles, R. R. J. Am. Chem. Soc. 2019, 141, 1457.
doi: 10.1021/jacs.8b12552 |
| [96] |
Zhao, K.; Yamashita, K.; Carpenter, J. E.; Sherwood, T. C.; Ewing, W. R.; Cheng, P. T. W.; Knowles, R. R. J. Am. Chem. Soc. 2019, 141, 8752.
doi: 10.1021/jacs.9b03973 pmid: 31117664 |
| [97] |
Nguyen, S. T.; Murray, P. R. D.; Knowles, R. R. ACS Catal. 2019, 10, 800.
doi: 10.1021/acscatal.9b04813 |
| [98] |
Huang, L.; Ji, T.; Rueping, M. J. Am. Chem. Soc. 2020, 142, 3532.
doi: 10.1021/jacs.9b12490 pmid: 32017543 |
| [99] |
Tsui, E.; Metrano, A. J.; Tsuchiya, Y.; Knowles, R. R. Angew. Chem., Int. Ed. 2020, 59, 11845.
doi: 10.1002/anie.v59.29 |
| [100] |
Tang, W.; Zhang, X. Chem. Rev. 2003, 103, 3029.
doi: 10.1021/cr020049i |
| [101] |
Piou, T.; Rovis, T. Nature 2015, 527, 86.
doi: 10.1038/nature15691 |
| [102] |
Bayeh, L.; Le, P. Q.; Tambar, U. K. Nature 2017, 547, 196.
doi: 10.1038/nature22805 |
| [103] |
Koh, M. J.; Nguyen, T. T.; Lam, J. K.; Torker, S.; Hyvl, J.; Schrock, R. R.; Hoveyda, A. H. Nature 2017, 542, 80.
doi: 10.1038/nature21043 |
| [104] |
Alonso, F.; Beletskaya, I. P.; Yus, M. Chem. Rev. 2004, 104, 3079.
doi: 10.1021/cr0201068 |
| [105] |
Xi, Y.; Dong, B.; McClain, E. J.; Wang, Q.; Gregg, T. L.; Akhmedov, N. G.; Petersen, J. L.; Shi, X. Angew. Chem., Int. Ed. 2014, 53, 4657.
doi: 10.1002/anie.201310142 |
| [106] |
Hopkinson, M. N.; Tlahuext-Aca, A.; Glorius, F. Acc. Chem. Res. 2016, 49, 2261.
doi: 10.1021/acs.accounts.6b00351 |
| [107] |
Patel, M.; Saunthwal, R. K.; Verma, A. K. Acc. Chem. Res. 2017, 50, 240.
doi: 10.1021/acs.accounts.6b00449 |
| [108] |
Wang, J.; Zhang, S.; Xu, C.; Wojtas, L.; Akhmedov, N. G.; Chen, H.; Shi, X. Angew. Chem., Int. Ed. 2018, 57, 6915.
doi: 10.1002/anie.v57.23 |
| [109] |
Wang, H.; Li, Y.; Tang, Z.; Wang, S.; Zhang, H.; Cong, H.; Lei, A. ACS Catal. 2018, 8, 10599.
doi: 10.1021/acscatal.8b02617 |
| [110] |
Liu, Y.; Chen, X. L.; Li, X. Y.; Zhu, S. S.; Li, S. J.; Song, Y.; Qu, L. B.; Yu, B. J. Am. Chem. Soc. 2021, 143, 964.
doi: 10.1021/jacs.0c11138 |
| [111] |
Kuss-Petermann, M.; Wenger, O. S. J. Phys. Chem. Lett. 2013, 4, 2535.
doi: 10.1021/jz4012349 |
| [112] |
Griesbaum, K. Angew. Chem., Int. Ed. 1970, 9, 273.
doi: 10.1002/(ISSN)1521-3773 |
| [113] |
Heiba, E.-A. I.; Dessau, R. M. J. Org. Chem. 1967, 32, 3837.
doi: 10.1021/jo01287a025 |
| [114] |
Huyser, E. S.; Kellogg, R. M. J. Org. Chem. 1966, 31, 3366.
doi: 10.1021/jo01348a059 |
| [115] |
Zhao, R.; Lind, J.; Merenyi, G.; Eriksen, T. E. J. Am. Chem. Soc. 1994, 116, 12010.
doi: 10.1021/ja00105a048 |
| [116] |
Dang, H.-S.; Roberts, B. P. Tetrahedron Lett. 1999, 40, 8929.
doi: 10.1016/S0040-4039(99)01898-5 |
| [117] |
Fujisawa, H.; Hayakawa, Y.; Sasaki, Y.; Mukaiyama, T. Chem. Lett. 2001, 30, 632.
doi: 10.1246/cl.2001.632 |
| [118] |
Benati, L.; Leardini, R.; Minozzi, M.; Nanni, D.; Scialpi, R.; Spagnolo, P.; Strazzari, S.; Zanardi, G. Angew. Chem., Int. Ed. 2004, 43, 3598.
doi: 10.1002/(ISSN)1521-3773 |
| [119] |
Kemper, J.; Studer, A. Angew. Chem., Int. Ed. 2005, 44, 4914.
doi: 10.1002/(ISSN)1521-3773 |
| [120] |
Dénès, F.; Pichowicz, M.; Povie, G.; Renaud, P. Chem. Rev. 2014, 114 2587.
doi: 10.1021/cr400441m |
| [121] |
Qvortrup, K.; Rankic, D. A.; MacMillan, D. W. J. Am. Chem. Soc. 2014, 136, 626.
doi: 10.1021/ja411596q pmid: 24341523 |
| [122] |
Hager, D.; MacMillan, D. W. J. Am. Chem. Soc. 2014, 136, 16986.
doi: 10.1021/ja5102695 |
| [123] |
Tyson, E. L.; Niemeyer, Z. L.; Yoon, T. P. J. Org. Chem. 2014, 79, 1427.
doi: 10.1021/jo500031g |
| [124] |
He, W.-B.; Gao, L.-Q.; Chen, X.-J.; Wu, Z.-L.; Huang, Y.; Cao, Z.; Xu, X.-H.; He, W.-M. Chin. Chem. Lett. 2020, 31, 1895.
doi: 10.1016/j.cclet.2020.02.011 |
| [125] |
Li, J.; Gu, Z.; Zhao, X.; Qiao, B.; Jiang, Z. Chem. Commun. 2019, 55, 12916.
doi: 10.1039/C9CC07380A |
| [126] |
Liu, X.; Yin, Y.; Jiang, Z. Chem. Commun. 2019, 55, 11527.
doi: 10.1039/C9CC06249A |
| [127] |
Yang, H.; Wei, G.; Jiang, Z. ACS Catal. 2019, 9, 9599.
doi: 10.1021/acscatal.9b03567 |
| [128] |
Zeng, G.; Li, Y.; Qiao, B.; Zhao, X.; Jiang, Z. Chem. Commun. 2019, 55, 11362.
doi: 10.1039/C9CC05304B |
| [129] |
Shi, J.; Wei, W. Chin. J. Org. Chem. 2020, 40, 2170. (in Chinese)
doi: 10.6023/cjoc202000041 |
|
(时建伟, 魏伟, 有机化学, 2020, 40 2170.)
doi: 10.6023/cjoc202000041 |
|
| [130] |
Prasanna, R.; Guha, S.; Sekar, G. Org. Lett. 2019, 21, 2650.
doi: 10.1021/acs.orglett.9b00635 |
| [131] |
Kong, M.; Tan, Y.; Zhao, X.; Qiao, B.; Tan, C. H.; Cao, S.; Jiang, Z. J. Am. Chem. Soc. 2021, 143, 4024.
doi: 10.1021/jacs.1c01073 |
| [132] |
Li, J.; He, L.; Liu, X.; Cheng, X.; Li, G. Angew. Chem., Int. Ed. 2019, 58, 1759.
doi: 10.1002/anie.v58.6 |
| [133] |
Zimmerman, H. E. Acc. Chem. Res. 2012, 45, 164.
doi: 10.1021/ar2000698 |
| [134] |
Benkeser, R. A.; Kaiser, E. M. J. Am. Chem. Soc. 1963, 85, 2858.
doi: 10.1021/ja00901a047 |
| [135] |
Swenson, K. E.; Zemach, D.; Nanjundiah, C.; Kariv-Miller, E. J. Org. Chem. 1983, 48, 1777.
doi: 10.1021/jo00158a042 |
| [136] |
Chaussard, J.; Combellas, C.; Thiebault, A. Tetrahedron Lett. 1987, 28, 1173.
|
| [137] |
Zhou, F.; Jehoulet, C.; Bard, A. J. J. Am. Chem. Soc. 1992, 114, 11004.
doi: 10.1021/ja00053a072 |
| [138] |
Ishifune, M.; Yamashita, H.; Kera, Y.; Yamashita, N.; Hirata, K.; Murase, H.; Kashimura, S. Electrochim. Acta 2003, 48, 2405.
doi: 10.1016/S0013-4686(03)00259-7 |
| [139] |
Peters, B. K.; Rodriguez, K. X.; Reisberg, S. H.; Beil, S. B.; Hickey, D. P.; Kawamata, Y.; Collins, M.; Starr, J.; Chen, L.; Udyavara, S.; Klunder, K.; Gorey, T. J.; Anderson, S. L.; Neurock, M.; Minteer, S. D.; Baran, P. S. Science 2019, 363, 838.
doi: 10.1126/science.aav5606 pmid: 30792297 |
| [140] |
Qin, Y.; Lu, J.; Zou, Z.; Hong, H.; Li, Y.; Li, Y.; Chen, L.; Hu, J.; Huang, Y. Org. Chem. Front. 2020, 7, 1817.
doi: 10.1039/D0QO00547A |
| [141] |
Liu, X.; Liu, R.; Qiu, J.; Cheng, X.; Li, G. Angew. Chem., Int. Ed. 2020, 59, 13962.
doi: 10.1002/anie.v59.33 |
| [142] |
Rossolini, T.; Leitch, J. A.; Grainger, R.; Dixon, D. J. Org. Lett. 2018, 20, 6794.
doi: 10.1021/acs.orglett.8b02923 pmid: 30350662 |
| [143] |
Vasu, D.; Fuentes de Arriba, A. L.; Leitch, J. A.; de Gombert, A.; Dixon, D. J. Chem. Sci. 2019, 10, 3401.
doi: 10.1039/C8SC05164J |
| [144] |
Nicastri, M. C.; Lehnherr, D.; Lam, Y. H.; DiRocco, D. A.; Rovis, T. J. Am. Chem. Soc. 2020, 142, 987.
doi: 10.1021/jacs.9b10871 pmid: 31904228 |
| [145] |
Lehnherr, D.; Lam, Y. H.; Nicastri, M. C.; Liu, J.; Newman, J. A.; Regalado, E. L.; DiRocco, D. A.; Rovis, T. J. Am. Chem. Soc. 2020, 142, 468.
doi: 10.1021/jacs.9b10870 pmid: 31849221 |
| [146] |
Chen, M.; Zhao, X.; Yang, C.; Xia, W. Org. Lett. 2017, 19, 3807.
doi: 10.1021/acs.orglett.7b01677 |
| [147] |
Cao, K.; Tan, S. M.; Lee, R.; Yang, S.; Jia, H.; Zhao, X.; Qiao, B.; Jiang, Z. J. Am. Chem. Soc. 2019, 141, 5437.
doi: 10.1021/jacs.9b00286 |
| [148] |
Huang, B.; Li, Y.; Yang, C.; Xia, W. Chem. Commun. 2019, 55, 6731.
doi: 10.1039/C9CC02368B |
| [149] |
Nageswar Rao, D.; Rasheed, S.; Raina, G.; Ahmed, Q. N.; Jaladanki, C. K.; Bharatam, P. V.; Das, P. J. Org. Chem. 2017, 82, 7234.
doi: 10.1021/acs.joc.7b00808 |
| [150] |
Zhang, G.; Fu, L.; Chen, P.; Zou, J.; Liu, G. Org. Lett. 2019, 21, 5015.
doi: 10.1021/acs.orglett.9b01607 |
| [151] |
Li, H; Zhang, M. T. Angew. Chem., Int. Ed. 2016, 55, 13132.
doi: 10.1002/anie.201607176 |
| [152] |
Schrauben, J. N.; Cattaneo, M.; Day, T. C.; Tenderholt, A. L.; Mayer, J. M. J. Am. Chem. Soc. 2012, 134, 16635.
doi: 10.1021/ja305668h pmid: 22974135 |
| [153] |
Sirimanne, C. T.; Kerrigan, M. M.; Martin, P. D.; Kanjolia, R. K.; Elliott, S. D.; Winter, C. H. Inorg. Chem. 2015, 54, 7.
doi: 10.1021/ic502184f pmid: 25488657 |
| [154] |
Canteenwala, T.; Padmawar, P. A.; Chiang, L. Y. J. Am. Chem. Soc. 2005, 127, 26.
pmid: 15631431 |
| [155] |
Irebo, T.; Reece, S. Y.; Sjödin, M.; Nocera, D. G.; Hammarström, L. J. Am. Chem. Soc. 2007, 129, 15462.
doi: 10.1021/ja073012u |
| [156] |
Pizano, A. A.; Yang, J. L.; Nocera, D. G. Chem. Sci. 2012, 3, 2457.
doi: 10.1039/c2sc20113e |
| [157] |
Kuss-Petermann, M.; Wolf, H.; Stalke, D.; Wenger, O. S. J. Am. Chem. Soc. 2012, 134, 12844.
doi: 10.1021/ja3053046 pmid: 22809316 |
| [158] |
Kuss-Petermann, M.; Wenger, O. S. J. Phys. Chem. A 2013, 117, 5726.
doi: 10.1021/jp402567m pmid: 23834357 |
| [159] |
Chen, J.; Kuss-Petermann, M.; Wenger, O. S. Chem.-Eur. J. 2014, 20, 4098.
doi: 10.1002/chem.201304256 |
| [160] |
Bronner, C.; Wenger, O. S. Phys. Chem. Chem. Phys. 2014, 16 3617.
|
| [161] |
Bowring, M. A.; Bradshaw, L. R.; Parada, G. A.; Pollock, T. P.; Fernández-Terán, R. J.; Kolmar, S. S.; Mercado, B. Q.; Schlenker, C. W.; Gamelin, D. R.; Mayer, J. M. J. Am. Chem. Soc. 2018, 140, 7449.
doi: 10.1021/jacs.8b04455 pmid: 29847111 |
| [162] |
Sayfutyarova, E. R.; Hammes-Schiffer, S. J. Phys. Chem. Lett. 2020, 11, 7109.
doi: 10.1021/acs.jpclett.0c02012 pmid: 32787327 |
| [163] |
Eisenhart, T. T.; Dempsey, J. L. J. Am. Chem. Soc. 2014, 136, 12221.
doi: 10.1021/ja505755k pmid: 25046022 |
| [164] |
Swords, W. B.; Meyer, G. J.; Hammarström, L. Chem. Sci. 2020, 11, 3460.
doi: 10.1039/C9SC04941J |
| [165] |
Lennox, J. C.; Kurtz, D. A.; Huang, T.; Dempsey, J. L. ACS Energy Lett. 2017, 2, 1246.
doi: 10.1021/acsenergylett.7b00063 |
| [166] |
Parada, G. A.; Goldsmith, Z. K.; Kolmar, S.; Pettersson Rimgard, B.; Mercado, B. Q.; Hammarström, L.; Hammes-Schiffer, S.; Mayer, J. M. Science 2019, 364, 471.
doi: 10.1126/science.aaw4675 |
| [167] |
Guo, J. D.; Yang, X. L.; Chen, B.; Tung, C. H.; Wu, L. Z. Org. Lett. 2020, 22, 9627.
doi: 10.1021/acs.orglett.0c03665 |
| [168] |
Lei, T.; Liang, G.; Cheng, Y. Y.; Chen, B.; Tung, C. H.; Wu, L. Z. Org. Lett. 2020, 22, 5385.
doi: 10.1021/acs.orglett.0c01709 |
| [169] |
Wang, J. H.; Li, X. B.; Li, J.; Lei, T.; Wu, H. L.; Nan, X. L.; Tung, C. H.; Wu, L. Z. Chem. Commun. 2019, 55, 10376.
doi: 10.1039/C9CC05375A |
| [170] |
Wang, J. H.; Lei, T.; Nan, X. L.; Wu, H. L.; Li, X. B.; Chen, B.; Tung, C. H.; Wu, L. Z. Org. Lett. 2019, 21, 5581.
doi: 10.1021/acs.orglett.9b01910 |
| [171] |
Huang, C.; Wang, J. H.; Qiao, J.; Fan, X. W.; Chen, B.; Tung, C. H.; Wu, L. Z. J. Org. Chem. 2019, 84, 12904.
doi: 10.1021/acs.joc.9b01603 |
| [172] |
Li, H.; Guo, Y. H.; Wu, J. Y.; Zhang, M. T. Chem. Commun. 2019, 55, 3465.
doi: 10.1039/C9CC00354A |
| [173] |
Chen, J.; Chen, J.; Liu, Y.; Zheng, Y.; Zhu, Q.; Han, G.; Shen, J.-R. J. Phys. Chem. Lett. 2019, 10, 3240.
doi: 10.1021/acs.jpclett.9b00959 pmid: 31117681 |
| [174] |
Ren, X.; Wang, X.; Sun, Y.; Chi, X.; Mangel, D.; Wang, H.; Sessler, J. L. Org. Chem. Front. 2019, 6, 584.
doi: 10.1039/C8QO01408F |
| [175] |
Lin, Y.; Yan, Y.; Peng, W.; Qiao, X.; Huang, D.; Ji, H.; Chen, C.; Ma, W.; Zhao, J. J. Phys. Chem. Lett. 2020, 11, 3941.
doi: 10.1021/acs.jpclett.0c01196 |
| [176] |
Zhang, X.; Ma, J.; Li, S.; Li, M.-D.; Guan, X.; Lan, X.; Zhu, R.; Phillips, D. L. J. Org. Chem. 2016, 81, 5330.
doi: 10.1021/acs.joc.6b00620 |
| [177] |
Xu, Y.; Bao, P.; Song, K.; Shi, Q. J. Comput. Chem. 2019, 40, 1005.
doi: 10.1002/jcc.v40.9 |
| [178] |
Zhong, W.; Wu, L.; Jiang, W.; Li, Y.; Mookan, N.; Liu, X. Dalton Trans. 2019, 48, 13711.
doi: 10.1039/c9dt02058f pmid: 31469134 |
| [179] |
Giret, Y.; Guo, P.; Wang, L.-F.; Cheng, J. J. Chem. Phys. 2020, 152, 124705.
doi: 10.1063/5.0001825 |
| [180] |
Song, K.; Shi, Q. J. Chem. Phys. 2017, 146, 184108.
doi: 10.1063/1.4982928 |
| [1] | Yatong Fu, Chaofan Sun, Dan Zhang, Chengguo Jin, Juyou Lu. Recent Progress in B—H Bond Functionalization of nido-Carboranes [J]. Chinese Journal of Organic Chemistry, 2024, 44(2): 438-447. |
| [2] | Jian Zhang, Wanjie Liang, Yi Yang, Fachao Yan, Hui Liu. Regiocontrollable Difunctionalization of N-Allenamines [J]. Chinese Journal of Organic Chemistry, 2024, 44(2): 335-348. |
| [3] | Luyao Li, Zhongwen He, Zhenguo Zhang, Zhenhua Jia, Teck-Peng Loh. Application of Triaryl Carbenium in Organic Synthesis [J]. Chinese Journal of Organic Chemistry, 2024, 44(2): 421-437. |
| [4] | Yanshuo Zhu, Hongyan Wang, Penghua Shu, Ke'na Zhang, Qilin Wang. Recent Advances on Alkoxy Radicals-Mediated C(sp3)—H Bond Functionalization via 1,5-Hydrogen Atom Transfer [J]. Chinese Journal of Organic Chemistry, 2024, 44(1): 1-17. |
| [5] | Jianghu Dong, Liangming Xuan, Chi Wang, Chenxi Zhao, Haifeng Wang, Qiongjiao Yan, Wei Wang, Fen'er Chen. Recent Advances in Visible-Light-Induced C(3)—H Functionalization of Quinoxalinones under Transition-Metal-Free or Photocatalyst-Free [J]. Chinese Journal of Organic Chemistry, 2024, 44(1): 111-136. |
| [6] | Qianfan Zhao, Yongzheng Chen, Shiming Zhang. Application and Mechanism Study of Carbon-Based Metal-Free Catalysts in Organic Synthesis [J]. Chinese Journal of Organic Chemistry, 2024, 44(1): 137-147. |
| [7] | Ran Zhou, Chunmei Yuan, Tao Zhang, Piao Mao, Yi Liu, Kaini Meng, Hui Xin, Wei Xue. Design, Synthesis and Bioactivity of Chalcone Derivative Containing Quinazolinone [J]. Chinese Journal of Organic Chemistry, 2023, 43(9): 3196-3209. |
| [8] | Yingjie Liu, Gangqing Shi, Ge Chou, Xin Zhang, Dongxue Song, Ning Chen, Miao Yu, Ying Xu. Progress of α-Position Functionalization of Ethers under Photo/Electrocatalysis [J]. Chinese Journal of Organic Chemistry, 2023, 43(8): 2664-2681. |
| [9] | Yuchao Wang, Jinbiao Liu, Zhitao He. Palladium-Catalyzed Asymmetric Hydrofunctionalizations of Conjugated Dienes [J]. Chinese Journal of Organic Chemistry, 2023, 43(8): 2614-2627. |
| [10] | Xiaoping Xu, Yifei Zhang, Xiaoyu Mo, Jun Jiang. Rh-Catalyzed C—H Functionalization Reaction between 3-Diazoindolin-2-imines and Pyrazolones for the Construction of 3-Pyrazolyl Indoles [J]. Chinese Journal of Organic Chemistry, 2023, 43(7): 2519-2527. |
| [11] | Rongbin Cai, Bing Li, Qi Zhou, Longyi Zhu, Jun Luo. Synthesis of 4,8,9,10-Tetrafunctionalized 2-Azaadamantanes and Their 2-Azaprotoadamantane Skeleton Isomers [J]. Chinese Journal of Organic Chemistry, 2023, 43(6): 2217-2225. |
| [12] | Jiamin Ma, Jiaoxiong Li, Qiansen Meng, Xianghua Zeng. Advances on the Radical Sulfonation of Alkynes [J]. Chinese Journal of Organic Chemistry, 2023, 43(6): 2040-2052. |
| [13] | Guangli Xu, Jing Xu, Haidong Xu, Xiang Cui, Xingzhong Shu. Research Progress of Transition Metal Catalyzed Synthesis of 1,3- Conjugated Diene Compounds from Alkenes and Alkynes [J]. Chinese Journal of Organic Chemistry, 2023, 43(6): 1899-1933. |
| [14] | Qian Dou, Taimin Wang, Lijing Fang, Hongbin Zhai, Bin Cheng. Recent Development of Photoinduced Iron-Catalysis in Organic Synthesis [J]. Chinese Journal of Organic Chemistry, 2023, 43(4): 1386-1415. |
| [15] | Shiquan Gao, Chuangjun Liu, Junfeng Yang, Junliang Zhang. Cobalt-Catalyzed Electrochemical Reductive Coupling of Alkynes and Alkenes [J]. Chinese Journal of Organic Chemistry, 2023, 43(4): 1559-1565. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||