化学学报 ›› 2026, Vol. 84 ›› Issue (5): 631-637.DOI: 10.6023/A25120420 上一篇 下一篇
研究论文
王炙韬a,†, 林雨珂a,†, 万妍a, 黄鑫伟a, 李云斌a, 吉定豪b,*( ), 项生昌a,*(
), 项生昌a,*( ), 张章静a,*(
), 张章静a,*( )
)
投稿日期:2025-12-25
发布日期:2026-02-28
通讯作者:
吉定豪, 项生昌, 张章静
作者简介:† 共同第一作者
★“框架材料化学”专辑
基金资助:
Zhitao Wanga, Yuke Lina, Yan Wana, Xinwei Huanga, Yunbin Lia, Dinghao Jib,*( ), Shengchang Xianga,*(
), Shengchang Xianga,*( ), Zhangjing Zhanga,*(
), Zhangjing Zhanga,*( )
)
Received:2025-12-25
Published:2026-02-28
Contact:
Dinghao Ji, Shengchang Xiang, Zhangjing Zhang
About author:† These authors contributed equally to this work
★ For the VSI “Chemistry of Framework Materials”.
Supported by:文章分享
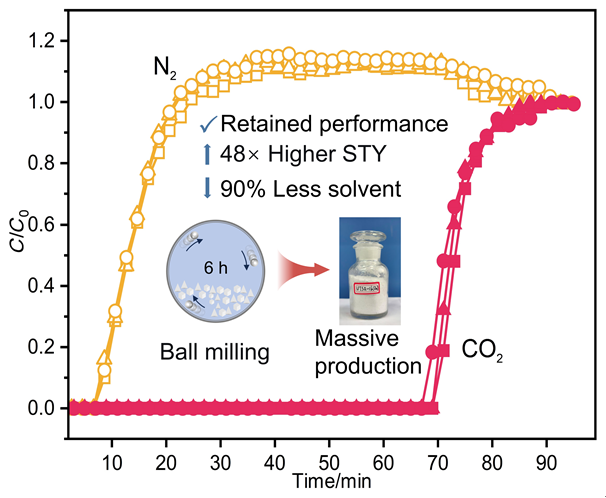
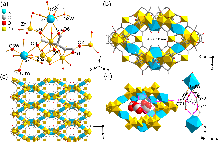
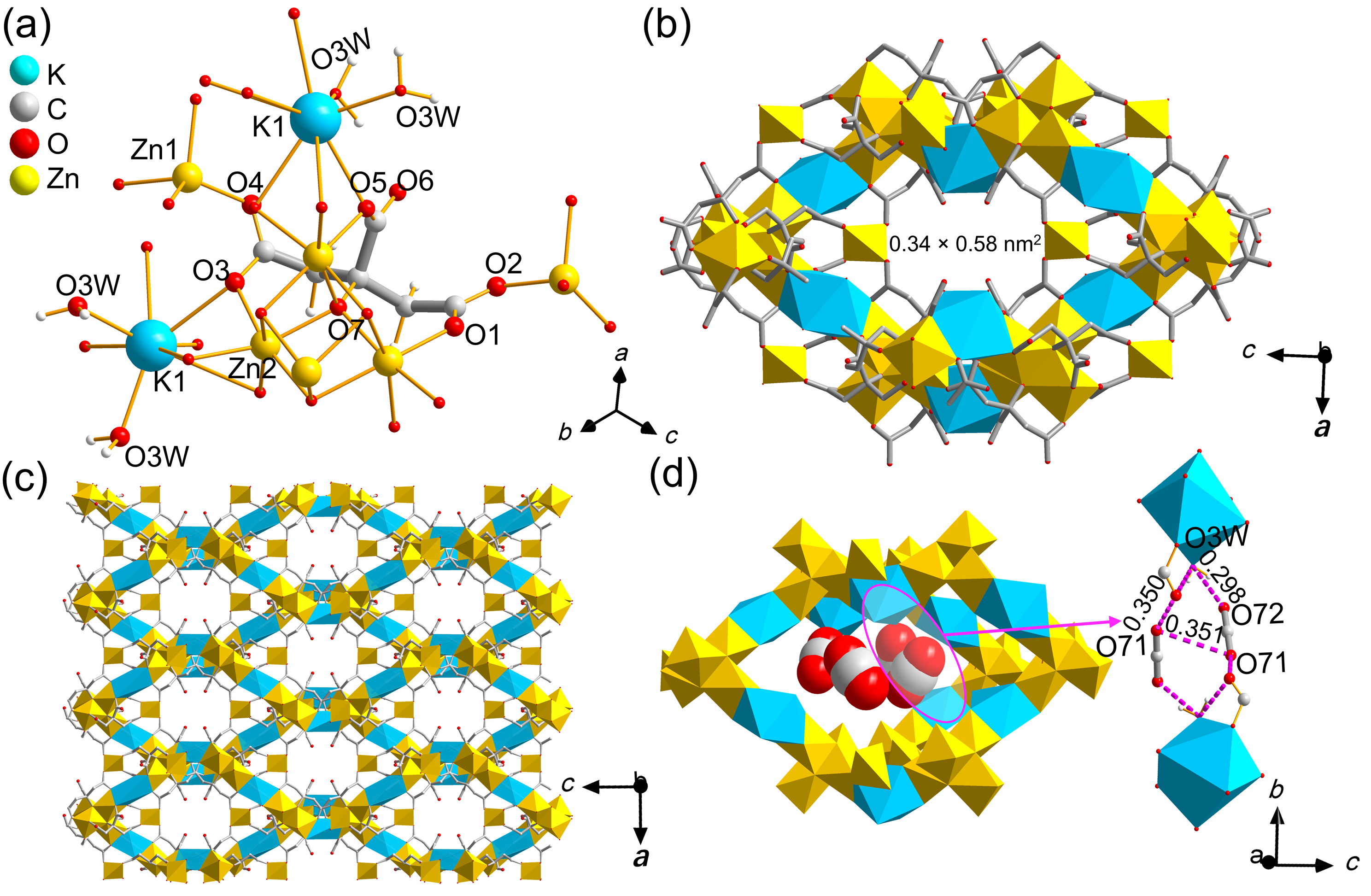
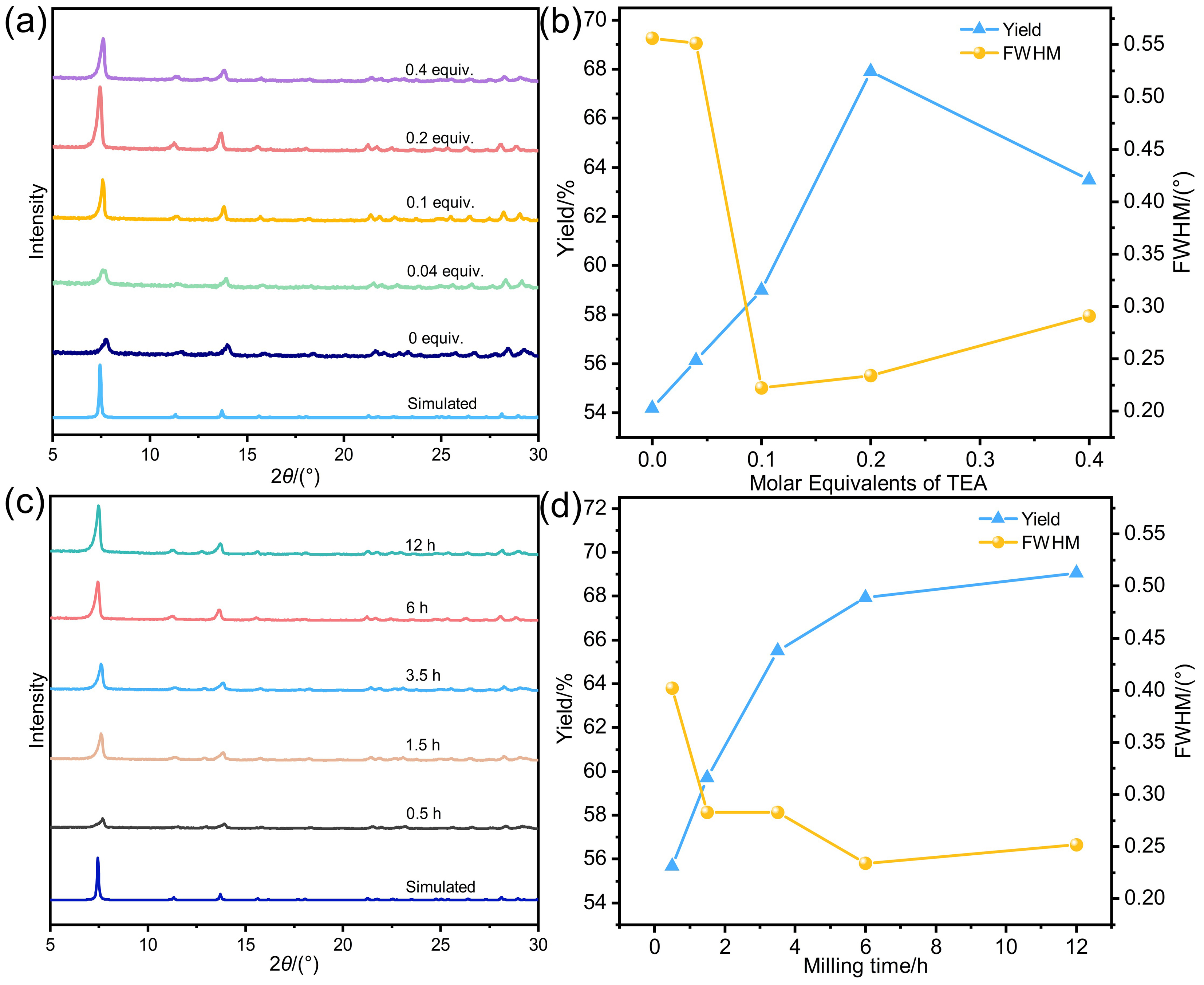
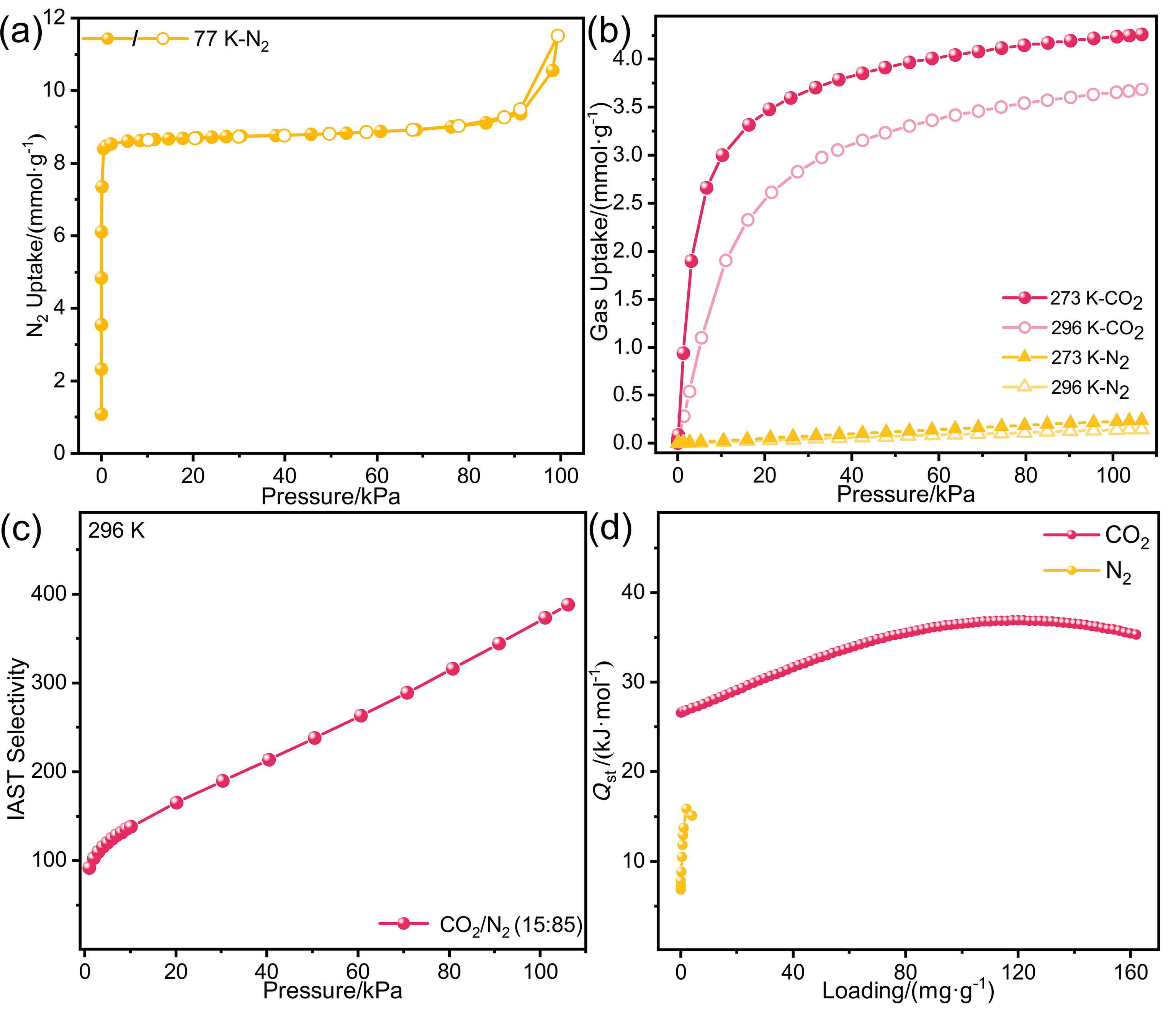
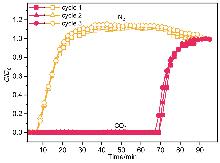
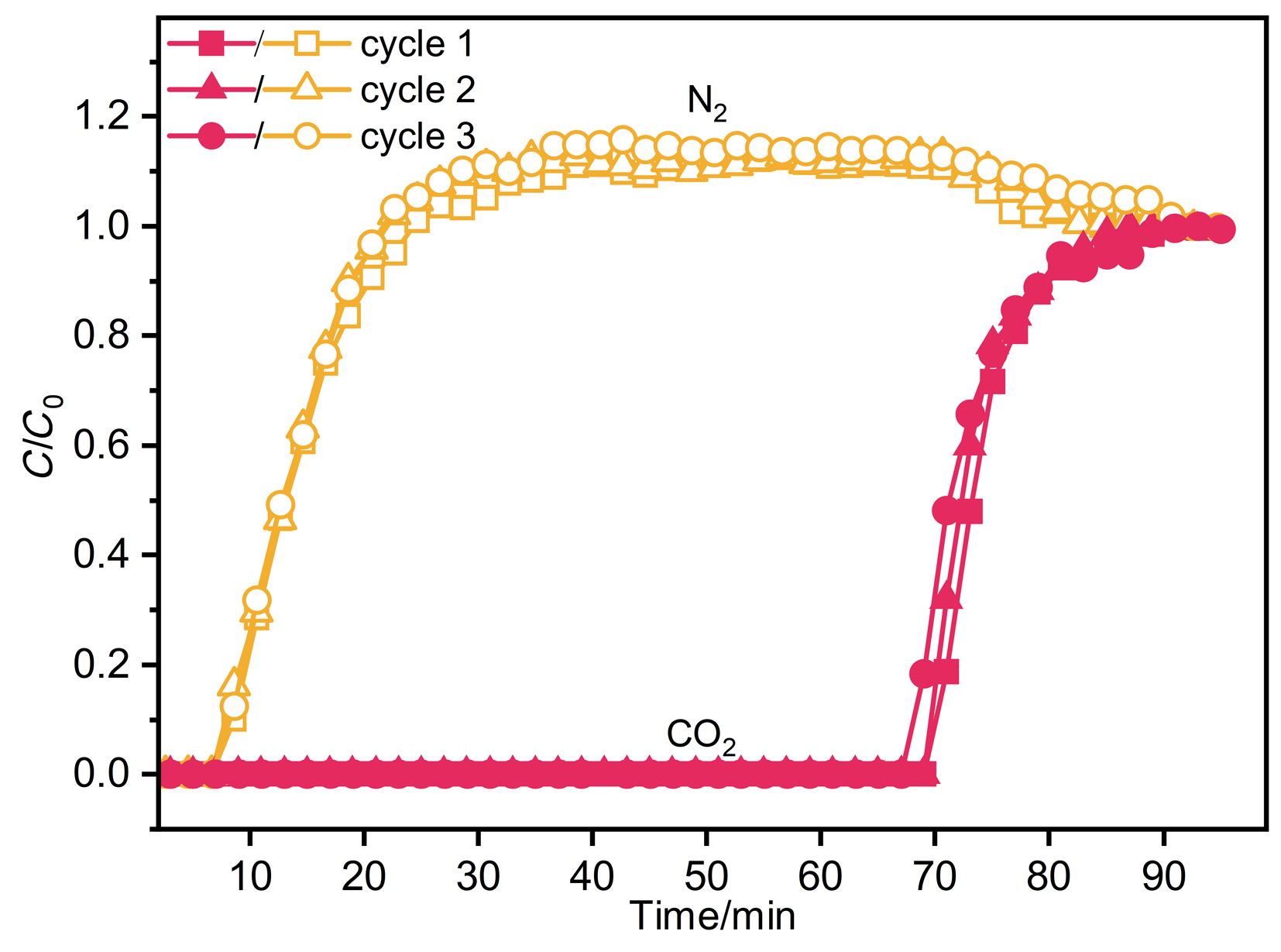
针对金属有机框架(MOFs)在工业化应用中面临的高能耗与大量溶剂浪费这一关键瓶颈, 本工作报道了一种快速、绿色的机械化学合成策略, 成功实现了高效CO2吸附剂UTSA-16(Zn)的宏量制备. 与传统溶剂热法相比, 该工艺将合成时间从48 h缩短至6 h, 使时空产率提升了48倍, 同时减少了约90%的溶剂消耗. 研究发现, 引入三乙胺作为碱调节剂, 对于调控反应体系pH值、抑制缺陷形成及确保产物的高结晶度至关重要. 所得材料的结构与水热法样品高度一致(BET比表面积达817 m2/g), 并完好保留了其优异的CO2捕获性能. 在296 K下, 材料的CO2吸附量达3.68 mmol/g, 对CO2/N2混合气表现出388的超高IAST (Ideal Adsorbed Solution Theory)选择性. 动态穿透实验进一步证实了该材料在模拟烟气条件下的卓越分离能力与循环稳定性.
王炙韬, 林雨珂, 万妍, 黄鑫伟, 李云斌, 吉定豪, 项生昌, 张章静. 快速、绿色机械化学合成UTSA-16(Zn)用于高效碳捕获★[J]. 化学学报, 2026, 84(5): 631-637.
Zhitao Wang, Yuke Lin, Yan Wan, Xinwei Huang, Yunbin Li, Dinghao Ji, Shengchang Xiang, Zhangjing Zhang. Rapid and Green Mechanochemical Synthesis of UTSA-16(Zn) for High-Performance CO2 Capture★[J]. Acta Chimica Sinica, 2026, 84(5): 631-637.
| [1] |
doi: 10.1038/46248 |
| [2] |
doi: 10.1038/nature02311 |
| [3] |
doi: 10.1038/nchem.444 |
| [4] |
doi: 10.1039/B618320B |
| [5] |
doi: 10.6023/A25040117 |
|
(高春, 张松涛, 庞欢, 化学学报, 2025, 83, 962.)
doi: 10.6023/A25040117 |
|
| [6] |
doi: 10.1021/ja3055639 pmid: 22906112 |
| [7] |
doi: 10.1021/acs.accounts.1c00707 |
| [8] |
doi: 10.1021/acs.accounts.1c00328 |
| [9] |
doi: 10.1021/ja803783c |
| [10] |
pmid: 11799235 |
| [11] |
doi: 10.1021/jacs.9b13072 |
| [12] |
doi: 10.1021/jacs.5c05068 |
| [13] |
doi: 10.1021/ja904782h |
| [14] |
doi: 10.1126/science.ady2607 |
| [15] |
doi: 10.1039/D4QI03110E |
| [16] |
doi: 10.1016/j.ccr.2025.217032 |
| [17] |
doi: 10.1126/science.aam7232 |
| [18] |
doi: 10.1126/science.abn8418 |
| [19] |
doi: 10.1038/s41586-021-03627-8 |
| [20] |
doi: 10.1002/anie.v62.37 |
| [21] |
doi: 10.6023/A17040168 |
|
(张贺, 李国良, 张可刚, 廖春阳, 化学学报, 2017, 75, 841.)
doi: 10.6023/A17040168 |
|
| [22] |
doi: 10.1002/cjoc.v43.10 |
| [23] |
doi: 10.6023/A25040140 |
|
(吴子林, 张璐, 陈杨, 李晋平, 李立博, 化学学报, 2025, 83, 917.)
doi: 10.6023/A25040140 |
|
| [24] |
doi: 10.6023/A15080547 |
|
(黄刚, 陈玉贞, 江海龙, 化学学报, 2016, 74, 113.)
doi: 10.6023/A15080547 |
|
| [25] |
doi: 10.1016/j.chempr.2023.09.009 |
| [26] |
doi: 10.1002/cssc.v18.11 |
| [27] |
doi: 10.1002/cjoc.v43.9 |
| [28] |
doi: 10.6023/A23100459 |
|
(刘洋, 高丰琴, 马占营, 张引莉, 李午戊, 侯磊, 张小娟, 王尧宇, 化学学报, 2024, 82, 152.)
doi: 10.6023/A23100459 |
|
| [29] |
doi: 10.1002/advs.v9.6 |
| [30] |
doi: 10.1002/adma.v36.27 |
| [31] |
doi: 10.1021/ja407219k |
| [32] |
|
|
(王跃, 邹莹, 张元, 郑舒婕, 王恒宇, 刘天赋, 李仁富, 化学学报, 2025, 83, 45.)
doi: 10.6023/A24110333 |
|
| [33] |
doi: 10.1021/ja0546065 |
| [34] |
doi: 10.1038/ncomms1956 |
| [35] |
doi: 10.1016/j.jiec.2020.04.015 |
| [36] |
doi: 10.1021/acs.inorgchem.0c01226 |
| [37] |
doi: 10.1016/j.cej.2020.126166 |
| [38] |
doi: 10.1016/j.micromeso.2021.111233 |
| [39] |
doi: 10.1039/d4dt02852j pmid: 39665278 |
| [40] |
doi: 10.1039/D5TA03364K |
| [41] |
doi: 10.1016/j.jece.2024.114167 |
| [42] |
doi: 10.1021/ja402727d |
| [1] | 陈慧滢, 黄宁宇, 廖培钦. 基于银−碳键的新型金属有机框架用于高效光催化合成过氧化氢★[J]. 化学学报, 2026, 84(5): 626-630. |
| [2] | 刘璐, 赵砚珑, 白薛峰, 张鑫, 李建荣. 可宏量制备的层叠配位聚合物用于CH4/N2分离★[J]. 化学学报, 2026, 84(5): 638-642. |
| [3] | 丁宝童, 蔡俊凯, 郭绍杰, 段春迎. 基于拓扑固定的铜卤簇金属有机框架的稳定性与发光调控研究★[J]. 化学学报, 2026, 84(5): 682-688. |
| [4] | 张伟宏, 马亚男, 方翰, 薛东旭. 面向乙烯一步纯化的多组分金属有机框架: 金属配体导向合成与C2混合气高效分离★[J]. 化学学报, 2026, 84(5): 659-666. |
| [5] | 冯超越, 薛波, 车国强, 刘大欢. 共价交联策略强化CO2/C2H2高效分离用IL@MOF复合材料的稳定性★[J]. 化学学报, 2026, 84(5): 673-681. |
| [6] | 欧阳阳, 俱战锋, 王文经, 杜顺福, 袁大强. 低成本高稳定性K2Zn3[Fe(CN)6]2框架用于高效CO2/C2H2分离★[J]. 化学学报, 2026, 84(5): 651-658. |
| [7] | 何乾龙, 胡洁颖, 钟礼匡, 何军. 基于金属有机框架的中性介质硝酸盐电催化还原制氨: 设计策略与机理研究★[J]. 化学学报, 2026, 84(5): 775-804. |
| [8] | 邬佳悦, 王文伯, 谢玉龙, 方伟慧. 铝基金属有机框架材料的主客体化学研究进展★[J]. 化学学报, 2026, 84(5): 697-708. |
| [9] | 张亚, 周功兵, 孙为银. 基于含氮杂环配体的金属有机框架材料在电催化CO2还原中的应用与前景展望★[J]. 化学学报, 2026, 84(5): 755-774. |
| [10] | 彭天资, 沈嘉克, 郭淑雅, 夏潇潇, 李炜. 迁移学习预测金属/共价有机骨架材料内小分子气体自扩散系数研究[J]. 化学学报, 2026, 84(3): 305-315. |
| [11] | 宋玉朋, 华紫辉, 葛介超, 王春儒, 吴波. 具有强大内建电场的C60@TpPa共价有机框架材料高效光催化产氢[J]. 化学学报, 2026, 84(3): 370-376. |
| [12] | 叶舣, 黄正义, 赵兴雷, 赵娅俐, 刘龙杰, 吴武凤, 魏嫣莹. 垂直排列金属有机框架纳米片膜实现高效H2传输[J]. 化学学报, 2026, 84(1): 129-134. |
| [13] | 税子怡, 尹书睿, 邓锦涛, 许留云, 郭莉. 聚乙烯吡咯烷酮辅助金属有机框架衍生分级多孔碳材料的制备及其双功能催化性能研究[J]. 化学学报, 2026, 84(1): 53-63. |
| [14] | 翁惠琼, 黄河, 王雯菲, 李和国, 李晓鹏, 张守鑫, 李树华, 赵越, 吴玉芳, 乔智威. 基于MOFid赋能下的AI大数据挖掘高性能化学战剂吸附材料[J]. 化学学报, 2026, 84(1): 8-19. |
| [15] | 江惠星, 郑丽彬, 陈小丰, 张珍珍, 陈秋水, 杨黄浩. 基于钛基金属有机框架的孔径调控策略及其在海水提铀中的应用★[J]. 化学学报, 2025, 83(9): 987-992. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||