有机化学 ›› 2026, Vol. 46 ›› Issue (3): 974-985.DOI: 10.6023/cjoc202508011 上一篇 下一篇
研究论文
周可a,†, 王泽涌a,†, 黎亚琳b, 张倩b, 李馨月b, 邹汉芳b, 张明忠a,*( ), 郭圣荣a,*(
), 郭圣荣a,*( ), 陈铁桥c
), 陈铁桥c
收稿日期:2025-08-08
修回日期:2025-11-27
发布日期:2025-12-25
作者简介:†共同第一作者
基金资助:
Ke Zhoua, Zeyong Wanga, Yalin Lib, Qian Zhangb, Xinyue Lib, Hanfang Zoub, Mingzhong Zhanga,*( ), Shengrong Guoa,*(
), Shengrong Guoa,*( ), Tieqiao Chenc
), Tieqiao Chenc
Received:2025-08-08
Revised:2025-11-27
Published:2025-12-25
Contact:
*E-mail: About author:†These authors contributed equally to this work.
Supported by:文章分享
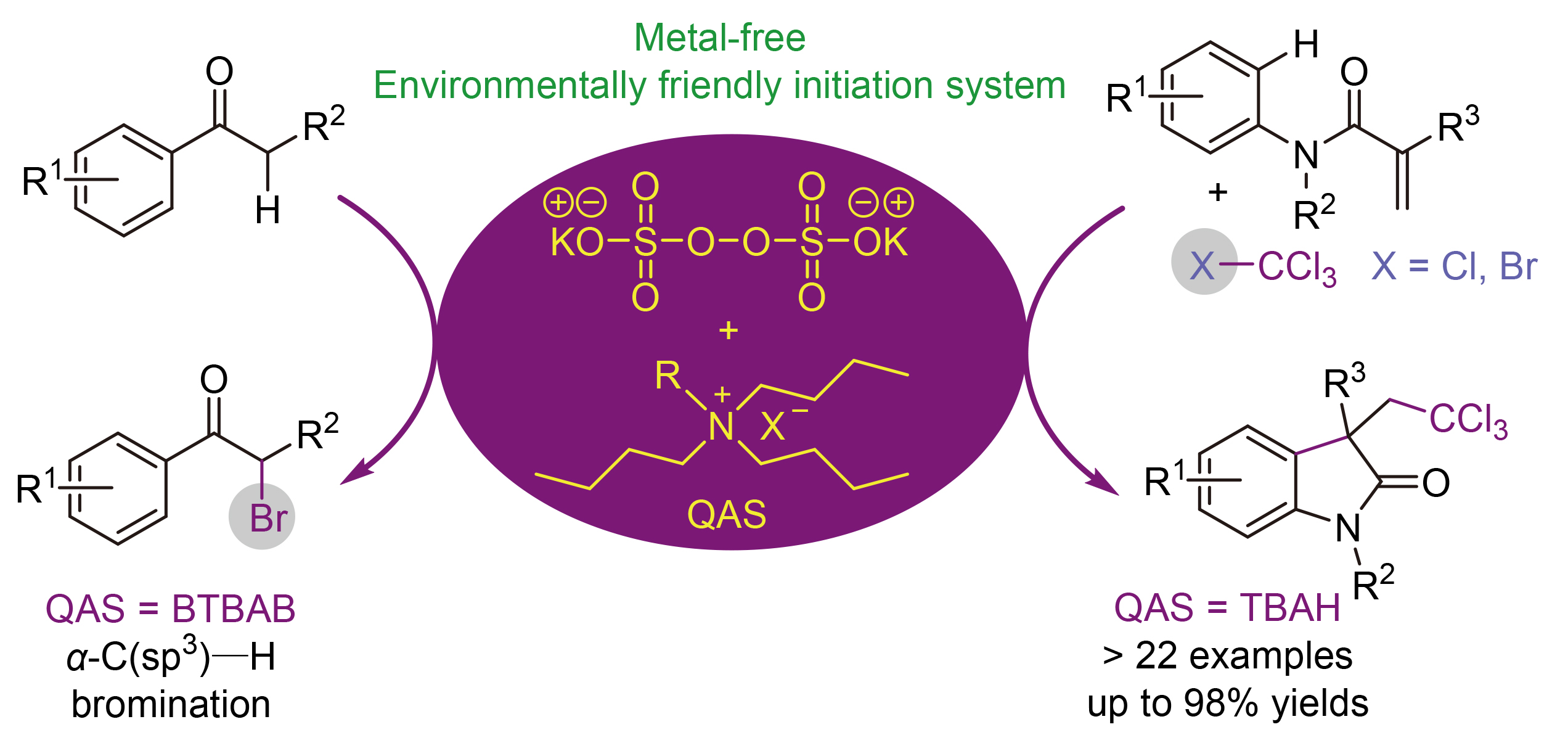
报道了一种由四卤化碳的C—X键断裂生成三氯甲基自由基的新型氧化方案. 该反应采用过硫酸钾(K2S2O8)与季铵盐(QAS)的组合作为自由基引发体系, 可在无过渡金属、无有机过氧化物条件下, 通过烯烃的级联环化反应合成三氯甲基化吲哚酮. 该方案能以中等到优异的收率得到目标产物, 且具有良好的官能团兼容性. 此外, 该催化体系还可用于酮的氧化C(sp³)—H键断裂反应, 实现由酮直接合成α-溴代酮.
周可, 王泽涌, 黎亚琳, 张倩, 李馨月, 邹汉芳, 张明忠, 郭圣荣, 陈铁桥. 一种由四卤化碳生成三氯甲基自由基的氧化策略: 无过渡金属合成三氯甲基化吲哚酮[J]. 有机化学, 2026, 46(3): 974-985.
Ke Zhou, Zeyong Wang, Yalin Li, Qian Zhang, Xinyue Li, Hanfang Zou, Mingzhong Zhang, Shengrong Guo, Tieqiao Chen. An Oxidative Strategy for Generating Trichloromethyl Radical from Carbon Tetrahalides: Transition-Metal-Free Synthesis of Trichloromethylated Oxindoles[J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 974-985.
| Entry | Oxidant (equiv.) | QAS (equiv.) | Yieldb/% |
|---|---|---|---|
| 1 | K2S2O8 (2.0) | TBAI (n-Bu4NI, 2.0) | 45 |
| 2 | K2S2O8 (2.0) | None | 0 |
| 3 | None | TBAI (n-Bu4NI, 2.0) | 0 |
| 4 | K2S2O8 (2.0) | TBAHS (n-Bu4NHSO4, 2.0) | 47 |
| 5 | K2S2O8 (2.0) | TBAB (n-Bu4NBr, 2.0) | 10 |
| 6 | K2S2O8 (2.0) | BTBAB (n-Bu3NBnBr, 2.0) | 13 |
| 7 | K2S2O8 (2.0) | TBAF (n-Bu4NF, 2.0) | 65 |
| 8 | K2S2O8 (2.0) | TBAH (n-Bu4NOH, 2.0) | 73 |
| 9 | K2S2O8 (2.0) | TBAH (n-Bu4NOH, 1.5) | 80 |
| 10 | K2S2O8 (2.0) | TBAH (n-Bu4NOH, 2.5) | 85 |
| 11 | K2S2O8 (2.5) | TBAH (n-Bu4NOH, 2.5) | 92 |
| 12 | K2S2O8 (3.0) | TBAH (n-Bu4NOH, 2.5) | 95 |
| 13c | K2S2O8 (3.0) | TBAH (n-Bu4NOH, 2.5) | 83 |
| 14d | K2S2O8 (3.0) | TBAH (n-Bu4NOH, 2.5) | 91 |
| Entry | Oxidant (equiv.) | QAS (equiv.) | Yieldb/% |
|---|---|---|---|
| 1 | K2S2O8 (2.0) | TBAI (n-Bu4NI, 2.0) | 45 |
| 2 | K2S2O8 (2.0) | None | 0 |
| 3 | None | TBAI (n-Bu4NI, 2.0) | 0 |
| 4 | K2S2O8 (2.0) | TBAHS (n-Bu4NHSO4, 2.0) | 47 |
| 5 | K2S2O8 (2.0) | TBAB (n-Bu4NBr, 2.0) | 10 |
| 6 | K2S2O8 (2.0) | BTBAB (n-Bu3NBnBr, 2.0) | 13 |
| 7 | K2S2O8 (2.0) | TBAF (n-Bu4NF, 2.0) | 65 |
| 8 | K2S2O8 (2.0) | TBAH (n-Bu4NOH, 2.0) | 73 |
| 9 | K2S2O8 (2.0) | TBAH (n-Bu4NOH, 1.5) | 80 |
| 10 | K2S2O8 (2.0) | TBAH (n-Bu4NOH, 2.5) | 85 |
| 11 | K2S2O8 (2.5) | TBAH (n-Bu4NOH, 2.5) | 92 |
| 12 | K2S2O8 (3.0) | TBAH (n-Bu4NOH, 2.5) | 95 |
| 13c | K2S2O8 (3.0) | TBAH (n-Bu4NOH, 2.5) | 83 |
| 14d | K2S2O8 (3.0) | TBAH (n-Bu4NOH, 2.5) | 91 |
| Entry | Oxidant (equiv.) | Additive (equiv.) | Solvent | Yieldb/% |
|---|---|---|---|---|
| 1 | K2S2O8 (2.0) | BTBAB (2.0) | PhCF3 | 78 |
| 2c | K2S2O8 (2.0) | BTBAB (2.0) | PhCF3 | 65 |
| 3 | K2S2O8 (2.0) | BTBAB (2.0) | DMF | 0 |
| 4 | K2S2O8 (2.0) | BTBAB (2.0) | MeCN | Trace |
| 5d | K2S2O8 (2.0) | BTBAB (2.0) | MeCN/H2O | 0 |
| 6 | K2S2O8 (2.0) | BTBAB (2.0) | MeNO2 | 19 |
| 7 | K2S2O8 (2.0) | BTBAB (1.5) | PhCF3 | 71 |
| 8 | K2S2O8 (2.0) | BTBAB (2.5) | PhCF3 | 79 |
| 9 | K2S2O8 (2.5) | BTBAB (2.0) | PhCF3 | 74 |
| 10 | K2S2O8 (2.0) | TBAB (2.0) | PhCF3 | 39 |
| 11 | K2S2O8 (2.0) | KBr (2.0) | PhCF3 | Trace |
| 12 | (NH4)2S2O8 (2.0) | BTBAB (2.0) | PhCF3 | 55 |
| 13 | Na2S2O8 (2.0) | BTBAB (2.0) | PhCF3 | 23 |
| Entry | Oxidant (equiv.) | Additive (equiv.) | Solvent | Yieldb/% |
|---|---|---|---|---|
| 1 | K2S2O8 (2.0) | BTBAB (2.0) | PhCF3 | 78 |
| 2c | K2S2O8 (2.0) | BTBAB (2.0) | PhCF3 | 65 |
| 3 | K2S2O8 (2.0) | BTBAB (2.0) | DMF | 0 |
| 4 | K2S2O8 (2.0) | BTBAB (2.0) | MeCN | Trace |
| 5d | K2S2O8 (2.0) | BTBAB (2.0) | MeCN/H2O | 0 |
| 6 | K2S2O8 (2.0) | BTBAB (2.0) | MeNO2 | 19 |
| 7 | K2S2O8 (2.0) | BTBAB (1.5) | PhCF3 | 71 |
| 8 | K2S2O8 (2.0) | BTBAB (2.5) | PhCF3 | 79 |
| 9 | K2S2O8 (2.5) | BTBAB (2.0) | PhCF3 | 74 |
| 10 | K2S2O8 (2.0) | TBAB (2.0) | PhCF3 | 39 |
| 11 | K2S2O8 (2.0) | KBr (2.0) | PhCF3 | Trace |
| 12 | (NH4)2S2O8 (2.0) | BTBAB (2.0) | PhCF3 | 55 |
| 13 | Na2S2O8 (2.0) | BTBAB (2.0) | PhCF3 | 23 |
| [1] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
|
(h)
|
|
| [2] |
|
| [3] |
(a)
|
|
(b)
|
|
| [4] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
|
(h)
|
|
| [5] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
| [6] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
|
(h)
|
|
| [7] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [8] |
(a)
|
|
(b)
|
|
| [9] |
|
| [10] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [11] |
(a)
|
|
(b)
|
|
| [12] |
|
| [13] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
| [14] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [15] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
|
(h)
|
|
|
(i)
|
|
|
(j)
|
|
|
(k)
|
|
|
(l)
|
|
|
(m)
|
|
|
(n)
|
|
| [16] |
For selected latest examples, see: (a) Wang, Q.-L.; Zhou, Q.; Liao, J.; Chen, Z.; Xiong, B.-Q.; Deng, G.-J.; Tang, K.-W.; Liu, Y. J. Org. Chem. 2021, 86, 2866.
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
|
(h)
|
|
|
(i)
|
|
|
(j)
|
|
| [17] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
| [18] |
|
| [19] |
An example of converting arylacrylamide with CCl4 into a trichloromethylated oxindole was also demonstrated in a metal-free phenol/Cs2CO3-promoted cascade cyclization of N-arylacrylamides with unactivated aryl and alkyl chlorides, see:
|
| [20] |
|
| [21] |
The α-bromination of ketones is a straightforward method for producing α-bromoketones, which are versatile intermediates in organic synthesis. Commonly used brominating systems in the reported reactions include liquid bromine (Br2), HBr/H2O2, CuBr₂, CuBr, NH4Br/H2SO4, NBS, dibromohydantoin (DBH), etc. However, these bromination systems exhibit at least one of the following drawbacks: (1) unsafe; (2) high cost and poor stability; (3) corrosive and inconvenient for transportation or storage; and ( 4) generation of deleterious waste which can increase the environmental burden. In contrast, this safe and environmentally friendly persulfate/bromide system demonstrates obvious advantages over the traditional brominating systems. For selected reactions using traditional brominating reagents mentioned above, see: (a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
|
(h)
|
|
|
(i)
|
|
|
(j)
|
|
|
(k)
|
|
|
(l)
|
|
|
(m)
|
|
| [22] |
The use of tetrabutylammonium tribromide (n-Bu4NBr3) as a source of Br2 in the α-bromination reaction is also documented in the literature, see:
|
| [23] |
|
| [24] |
More optimizations can be found in the Supporting Information.
|
| [25] |
|
| [26] |
|
| [27] |
(a)
|
|
(b)
|
| [1] | 印丘梅, 刘佳乐, 邓世强, 黄嘉明, 蒋洁, 唐裕才. 锰促进活化烯烃与亚磷酸酯串联磷酰化/环化反应合成γ-咔啉酮衍生物[J]. 有机化学, 2026, 46(3): 1078-1086. |
| [2] | 韦翠, 刘金秋, 邓灿, 邹宁, 周文俊. 螺芴基化合物的合成研究进展[J]. 有机化学, 2026, 46(3): 817-839. |
| [3] | 黄泽宁, 左凯丞, 胡涛, 张澄阳, 张金龙, 蒋高喜. 膦酸催化的双/三次Pictet-Spengler缩合/氧化芳构化构建C2和C3对称的喹啉酮[J]. 有机化学, 2026, 46(3): 1070-1077. |
| [4] | 高源, 刘岩, 李师伍, 唐天胜, 孟钰坤, 赵志飞. 氮杂环卡宾(NHC)催化[3+2]环加成反应高非对映选择性地构建环戊酮-螺环氧化吲哚化合物[J]. 有机化学, 2026, 46(3): 1008-1016. |
| [5] | 孙雨, 朱延涛, 张东, 倪春节. 镍催化炔基与三氟乙酰亚胺氯和水的环化反应: 高效合成2-(三氟甲基)吲哚衍生物[J]. 有机化学, 2026, 46(3): 1017-1026. |
| [6] | 马超, 徐晓玲, 骆翔, 鲁瑞娟, 虞国棋, 蔡涛. 电化学促进邻炔基苯甲酸酯与硫酚的自由基串联环化反应合成4-硫代异香豆素[J]. 有机化学, 2026, 46(3): 1039-1049. |
| [7] | 王馨玉, 黄晨佩, 郭昌, 宋琎. 电化学驱动芳基膦氧化物自由基二烯基化反应[J]. 有机化学, 2026, 46(3): 1050-1059. |
| [8] | 杨其昌, 张筱睿, 吕剑, 古双喜. 糖羟基的选择性氧化反应研究进展[J]. 有机化学, 2026, 46(3): 743-758. |
| [9] | 夏春年, 陈凯林, 金诺琪, 南欣卉, 汪兵洋, 吴馨怡, 孙强盛, 孙伟. 仿生四齿胺基吡啶锰催化剂/H2O2的醇类化合物高效氧化: 间歇和连续流研究[J]. 有机化学, 2026, 46(3): 986-992. |
| [10] | 黄涎廷, 沈小倩, 王庆许, 柳忠全. 无催化剂及无溶剂光引发下氮杂芳烃的自由基脱羧酰基化反应[J]. 有机化学, 2026, 46(3): 951-960. |
| [11] | 何雨蒙, 周风院, 江楠, 张玉琦, 王记江, 马豪杰. 碘催化分子内环化2-羟基-吲哚-3-酮类化合物的合成[J]. 有机化学, 2026, 46(3): 961-967. |
| [12] | 杨利婷, 潘慧茹, 许晓旭, 张益恺, 来姝妤, 曹信誉, 刘国群, 乔辉杰, 焦明立. 一种无过渡金属、绿色温和的一锅法合成3-碘代/溴代咪唑并[1,2-a]吡啶类化合物[J]. 有机化学, 2026, 46(3): 915-924. |
| [13] | 刘琳, 周璐璐, 梁天宇, 钟克利, 燕小梅, 汤立军. 基于苯并吡喃衍生物的近红外荧光探针用于二氧化硫衍生物的高灵敏检测与生物成像[J]. 有机化学, 2026, 46(3): 866-871. |
| [14] | 李明, 王智超, 石大伟, 杨凤坤, 王长城, 杨斌. 亚化学计量三氟甲烷磺酸酐/二甲基亚砜协同促进的三价磷到三级磷氧化物的氧化转化[J]. 有机化学, 2026, 46(3): 888-897. |
| [15] | 李佳豪, 袁冰, 于凤丽. 碳@硅复合材料负载的五氧化二铌催化环戊烯环氧化反应研究[J]. 有机化学, 2026, 46(2): 624-632. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||