有机化学 ›› 2026, Vol. 46 ›› Issue (3): 915-924.DOI: 10.6023/cjoc202509015 上一篇 下一篇
研究论文
杨利婷, 潘慧茹, 许晓旭, 张益恺, 来姝妤, 曹信誉, 刘国群*( ), 乔辉杰*(
), 乔辉杰*( ), 焦明立*(
), 焦明立*( )
)
收稿日期:2025-09-11
修回日期:2025-12-04
发布日期:2026-01-06
基金资助:
Liting Yang, Huiru Pan, Xiaoxu Xu, Yikai Zhang, Shuyu Lai, Xinyu Cao, Guoqun Liu*( ), Huijie Qiao*(
), Huijie Qiao*( ), Mingli Jiao*(
), Mingli Jiao*( )
)
Received:2025-09-11
Revised:2025-12-04
Published:2026-01-06
Contact:
*E-mail: Supported by:文章分享
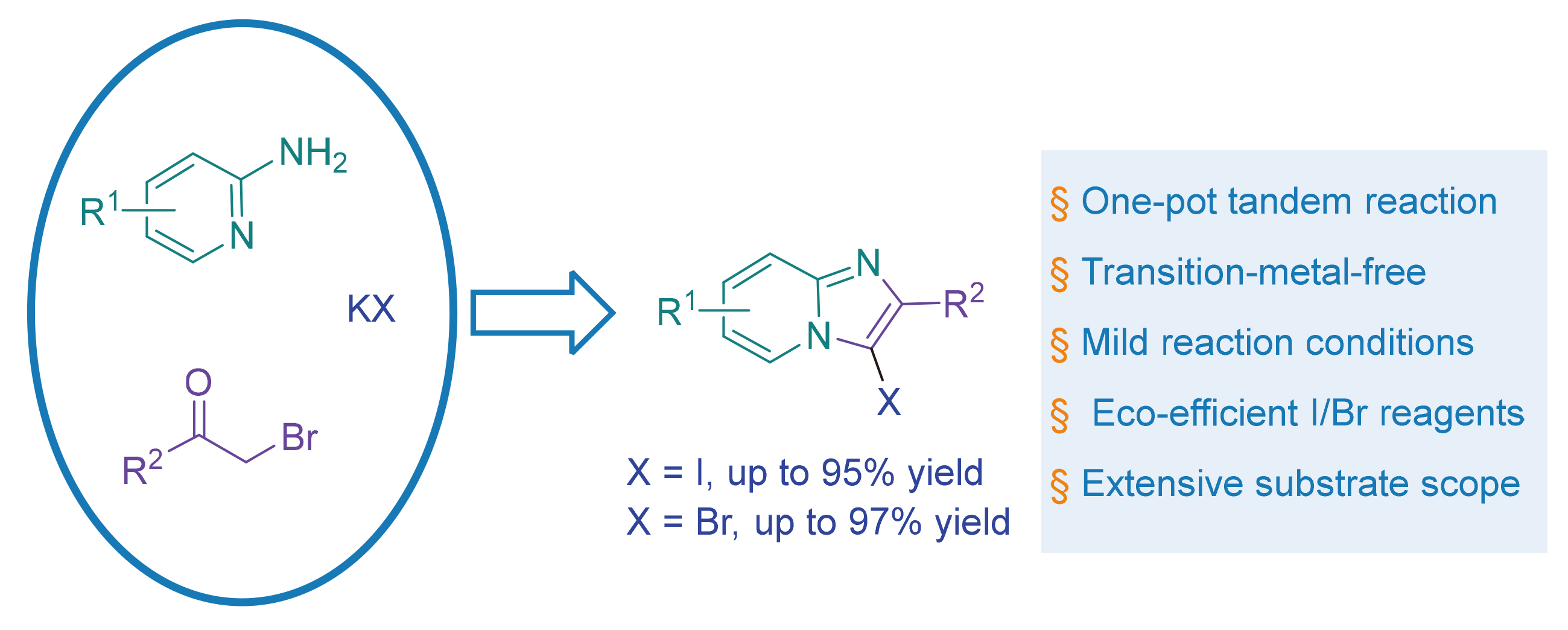
以2-氨基吡啶类化合物、甲酰基甲基溴类化合物和碘化钾(KI)为反应物, 建立了一种无过渡金属参与的一锅多步策略, 用于高效合成3-碘代咪唑并[1,2-a]吡啶衍生物. 该方法具有显著优势: 无需过渡金属、反应条件温和(在室温下进行), 且使用绿色无毒的碘化试剂. 值得注意的是, 带有给电子基团和吸电子基团的2-氨基吡啶与甲酰基甲基溴均表现出良好的兼容性, 其中带有给电子基团的底物通常能获得略高的产率. 通过对照实验进行机理研究表明, 该反应遵循两步反应机理: 首先, 2-氨基吡啶与甲酰基甲基溴反应生成咪唑并[1,2-a]吡啶中间体; 随后, 该中间体与KI发生C3位碘化反应. 此外, 该策略可扩展至3-溴代咪唑并[1,2-a]吡啶的合成. 在优化条件(65 ℃, 6 h)下, 以溴化钾(KBr)为溴源, 产物产率可达55%~97%.
杨利婷, 潘慧茹, 许晓旭, 张益恺, 来姝妤, 曹信誉, 刘国群, 乔辉杰, 焦明立. 一种无过渡金属、绿色温和的一锅法合成3-碘代/溴代咪唑并[1,2-a]吡啶类化合物[J]. 有机化学, 2026, 46(3): 915-924.
Liting Yang, Huiru Pan, Xiaoxu Xu, Yikai Zhang, Shuyu Lai, Xinyu Cao, Guoqun Liu, Huijie Qiao, Mingli Jiao. A Green and Mild One-Pot Protocol for the Synthesis of 3-Iodo-/Bromo-imidazo[1,2-a]pyridines without Transition Metals[J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 915-924.
| Entry | Solvent | Oxidant | Yieldb/% |
|---|---|---|---|
| 1 | DCE | K2S2O8 | 86 |
| 2 | PE | K2S2O8 | 48 |
| 3 | EA | K2S2O8 | <5 |
| 4 | DMF | K2S2O8 | <5 |
| 5 | 1,4-Dioxane | K2S2O8 | 63 |
| 6 | EtOH | K2S2O8 | 67 |
| 7 | Toluene | K2S2O8 | 55 |
| 8 | CH3CN | K2S2O8 | 23 |
| 9 | H2O | K2S2O8 | 76 |
| 10 | CH3CN/H2O (V/V=2/1) | K2S2O8 | 91 |
| 11 | CH3CN/H2O (V/V=1/2) | K2S2O8 | 88 |
| 12 | CH3CN/H2O (V/V=1/1) | K2S2O8 | 94 |
| 13 | CH3CN/H2O (V/V=1/1) | TBHP | <5 |
| 14 | CH3CN/H2O (V/V=1/1) | DTBP | <5 |
| 15 | CH3CN/H2O (V/V=1/1) | Na2S2O8 | 90 |
| 16 | CH3CN/H2O (V/V=1/1) | (NH4)2S2O8 | 78 |
| 17 | CH3CN/H2O (V/V=1/1) | KHSO5 | 82 |
| 18 | CH3CN/H2O (V/V=1/1) | — | <5 |
| 19c | CH3CN/H2O (V/V=1/1) | K2S2O8 | 94 |
| 20d | CH3CN/H2O (V/V=1/1) | K2S2O8 | 92 |
| Entry | Solvent | Oxidant | Yieldb/% |
|---|---|---|---|
| 1 | DCE | K2S2O8 | 86 |
| 2 | PE | K2S2O8 | 48 |
| 3 | EA | K2S2O8 | <5 |
| 4 | DMF | K2S2O8 | <5 |
| 5 | 1,4-Dioxane | K2S2O8 | 63 |
| 6 | EtOH | K2S2O8 | 67 |
| 7 | Toluene | K2S2O8 | 55 |
| 8 | CH3CN | K2S2O8 | 23 |
| 9 | H2O | K2S2O8 | 76 |
| 10 | CH3CN/H2O (V/V=2/1) | K2S2O8 | 91 |
| 11 | CH3CN/H2O (V/V=1/2) | K2S2O8 | 88 |
| 12 | CH3CN/H2O (V/V=1/1) | K2S2O8 | 94 |
| 13 | CH3CN/H2O (V/V=1/1) | TBHP | <5 |
| 14 | CH3CN/H2O (V/V=1/1) | DTBP | <5 |
| 15 | CH3CN/H2O (V/V=1/1) | Na2S2O8 | 90 |
| 16 | CH3CN/H2O (V/V=1/1) | (NH4)2S2O8 | 78 |
| 17 | CH3CN/H2O (V/V=1/1) | KHSO5 | 82 |
| 18 | CH3CN/H2O (V/V=1/1) | — | <5 |
| 19c | CH3CN/H2O (V/V=1/1) | K2S2O8 | 94 |
| 20d | CH3CN/H2O (V/V=1/1) | K2S2O8 | 92 |
| [1] |
doi: 10.1039/C4CC08495K |
| [2] |
doi: 10.1002/tcr.v24.12 |
| [3] |
|
|
(刘想, 李文, 刘环宇, 曹华, 有机化学, 2021, 41, 1759.)
doi: 10.6023/cjoc202012026 |
|
| [4] |
doi: 10.2165/00023210-200519010-00008 |
| [5] |
doi: 10.1016/j.pestbp.2021.104857 |
| [6] |
doi: 10.6023/cjoc201712045 |
|
(于文全, 宋利娜, 田显海, 赵婷, 王漫漫, 武杰, 乔艳, 常俊标, 有机化学, 2018, 38, 1530.)
doi: 10.6023/cjoc201712045 |
|
| [7] |
doi: 10.1016/j.bmc.2021.116362 |
| [8] |
doi: 10.1002/chem.v26.39 |
| [9] |
|
| [10] |
doi: 10.1021/acs.jmedchem.0c02163 |
| [11] |
doi: 10.1002/anie.v50.8 |
| [12] |
doi: 10.1039/c2jm14904d |
| [13] |
doi: 10.1016/j.tetlet.2024.155135 |
| [14] |
doi: 10.1002/cjoc.v40.19 |
| [15] |
doi: 10.1016/j.tetlet.2021.153028 |
| [16] |
doi: 10.1055/s-0035-1562492 |
| [17] |
doi: 10.1002/jhet.v57.11 |
| [18] |
doi: 10.1039/D2NJ00159D |
| [19] |
doi: 10.1039/D0GC00137F |
| [20] |
doi: 10.1002/asia.v20.2 |
| [21] |
doi: 10.1021/cr400215u |
| [22] |
doi: 10.1021/ar500101a |
| [23] |
doi: 10.1039/D1CS00399B |
| [24] |
doi: 10.1039/D1QO00313E |
| [25] |
doi: 10.1021/cr1002084 pmid: 21105733 |
| [26] |
doi: 10.6023/cjoc201612038 |
|
(陈亮, 王保取, 赵宇澄, 严胜骄, 林军, 有机化学, 2017, 37, 1433.)
doi: 10.6023/cjoc201612038 |
|
| [27] |
doi: 10.1016/j.jcat.2025.115938 |
| [28] |
doi: 10.6023/cjoc201711014 |
| [29] |
doi: 10.1016/j.cclet.2025.111402 |
| [30] |
doi: 10.1039/C4CY00919C |
| [31] |
doi: 10.1021/acs.langmuir.4c03052 |
| [32] |
doi: 10.1039/D4RA02090A |
| [33] |
doi: 10.1055/s-0037-1611856 |
| [34] |
doi: 10.1021/acs.joc.8b02637 |
| [35] |
doi: 10.1080/00397911.2020.1717539 |
| [36] |
doi: 10.1039/D3GC03069E |
| [37] |
doi: 10.6023/cjoc202110015 |
|
(乔辉杰, 杨利婷, 陈雅, 王嘉琳, 孙武轩, 董昊博, 王云威, 有机化学, 2022, 42, 1188.)
doi: 10.6023/cjoc202110015 |
|
| [38] |
doi: 10.1002/chem.v30.61 |
| [39] |
doi: 10.1021/acs.joc.4c00173 |
| [40] |
doi: 10.6023/cjoc201904068 |
|
(徐鑫明, 陈德茂, 王祖利, 有机化学, 2019, 39, 3338.)
doi: 10.6023/cjoc201904068 |
|
| [41] |
doi: 10.1002/ejoc.v2018.26 |
| [42] |
doi: 10.6023/cjoc202412022 |
|
(张连吉, 王炳福, 潘大龙, 王翠苹, 赵玉辉, 迟海军, 董岩, 魏万国, 张志强, 有机化学, 2025, 45, 2923.)
doi: 10.6023/cjoc202412022 |
|
| [43] |
doi: 10.6023/cjoc201904057 |
|
(王薪, 穆石强, 孙婷, 孙凯, 有机化学, 2019, 39, 2802.)
doi: 10.6023/cjoc201904057 |
|
| [44] |
doi: 10.1002/slct.v5.40 |
| [45] |
doi: 10.1021/acs.orglett.5b01868 |
| [46] |
|
|
(张周, 郭钰, 羊静, 吴丹, 王佳昕, 洪欣玥, 蔡佩君, 荣良策, 有机化学, 2023, 43, 2104.)
doi: 10.6023/cjoc202212014 |
|
| [47] |
doi: 10.2174/1570178619666220806111324 |
| [48] |
doi: 10.1021/acs.joc.2c00529 |
| [49] |
doi: 10.1002/cjoc.v37.6 |
| [1] | 周可, 王泽涌, 黎亚琳, 张倩, 李馨月, 邹汉芳, 张明忠, 郭圣荣, 陈铁桥. 一种由四卤化碳生成三氯甲基自由基的氧化策略: 无过渡金属合成三氯甲基化吲哚酮[J]. 有机化学, 2026, 46(3): 974-985. |
| [2] | 赵宇含, 马惠敏, 戈书林, 孔令斌. 碘催化烯烃与活性亚甲基化合物的分子间环丙烷化反应[J]. 有机化学, 2025, 45(9): 3469-3477. |
| [3] | 刘梦琴, 陈贻庭, 张榉元, 周和烨, 秦涛, 刘彬. 三组分交叉偶联反应高效合成双芳基硫醚[J]. 有机化学, 2025, 45(4): 1283-1296. |
| [4] | 谢桂玲, Sudhakar Reddy Mopuri, 李杰, 杨元勇, 陈铁根. 无过渡金属参与的“一锅法”合成咔啉及其衍生物的新方法[J]. 有机化学, 2025, 45(10): 3824-3837. |
| [5] | 黄真茹, 金国顺, 陈天煜, 冯斌, 史鑫康, 陈敏方, 华路生, 徐清. 氢氧化铯催化温和有氧环化反应高效构建喹喔啉杂环衍生物[J]. 有机化学, 2024, 44(9): 2933-2942. |
| [6] | 田永盛, 魏斓枫, 黄嘉为, 韦玉, 徐亮, 刘帅. 四丁基三溴化铵促进的有机硼酸在无过渡金属条件下的脱硼硒化、溴化和羟基化反应[J]. 有机化学, 2024, 44(6): 1987-1997. |
| [7] | 鞠国栋, 周冠宇, 赵应声. 三异丙基硅烷(TIPS)保护苯酚的无过渡金属催化区域选择性硫氰化反应[J]. 有机化学, 2024, 44(4): 1327-1336. |
| [8] | 万云辉, 杨福美, 陈明瀚, 孙德立, 叶丹锋. 无过渡金属催化的N-苄基-N-叔丁氧羰基酰胺与不饱和醇的酯化反应[J]. 有机化学, 2024, 44(4): 1293-1300. |
| [9] | 曹同阳, 李玮, 王力竞. N-碘代丁二酰亚胺(NIS)参与的碘化反应最新研究进展[J]. 有机化学, 2024, 44(2): 508-524. |
| [10] | 刘杰, 韩峰, 李双艳, 陈天煜, 陈建辉, 徐清. 无过渡金属参与甲基杂环化合物与醇的选择性有氧烯基化反应[J]. 有机化学, 2024, 44(2): 573-583. |
| [11] | 董江湖, 宣良明, 王池, 赵晨熙, 王海峰, 严琼姣, 汪伟, 陈芬儿. 无过渡金属或无光催化剂条件下可见光促进喹喔啉酮C(3)—H官能团化研究进展[J]. 有机化学, 2024, 44(1): 111-136. |
| [12] | 徐忠荣, 万结平, 刘云云. 基于热、光以及电化学过程的无过渡金属碳-氢键硫氰化和硒氰化反应[J]. 有机化学, 2023, 43(7): 2425-2446. |
| [13] | 秦娇, 陈杰, 苏艳. 无过渡金属催化的α-溴代茚酮自由基裂解反应合成(2-氰基苯基)乙酸-2,2,6,6-四甲基哌啶酯[J]. 有机化学, 2023, 43(6): 2171-2177. |
| [14] | 王睿, 高朗, 周岑, 张霄. 苯基吩噻嗪多孔有机聚合物催化的非活化末端烯烃的卤代全氟烷基化反应[J]. 有机化学, 2023, 43(3): 1136-1145. |
| [15] | 孙婧, 张萌萌, 锅小龙, 王琪, 王陆瑶. 无过渡金属条件下二芳基硒化合物的合成[J]. 有机化学, 2023, 43(12): 4251-4260. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||