有机化学 ›› 2026, Vol. 46 ›› Issue (3): 697-724.DOI: 10.6023/cjoc202508020 上一篇 下一篇
综述与进展
收稿日期:2025-08-23
修回日期:2025-11-14
发布日期:2025-12-09
基金资助:
Hui Luo, Wenquan Wang, Yu He, Fuqiang Wang*( ), Jinhui Yang*(
), Jinhui Yang*( )
)
Received:2025-08-23
Revised:2025-11-14
Published:2025-12-09
Contact:
*E-mail: Supported by:文章分享
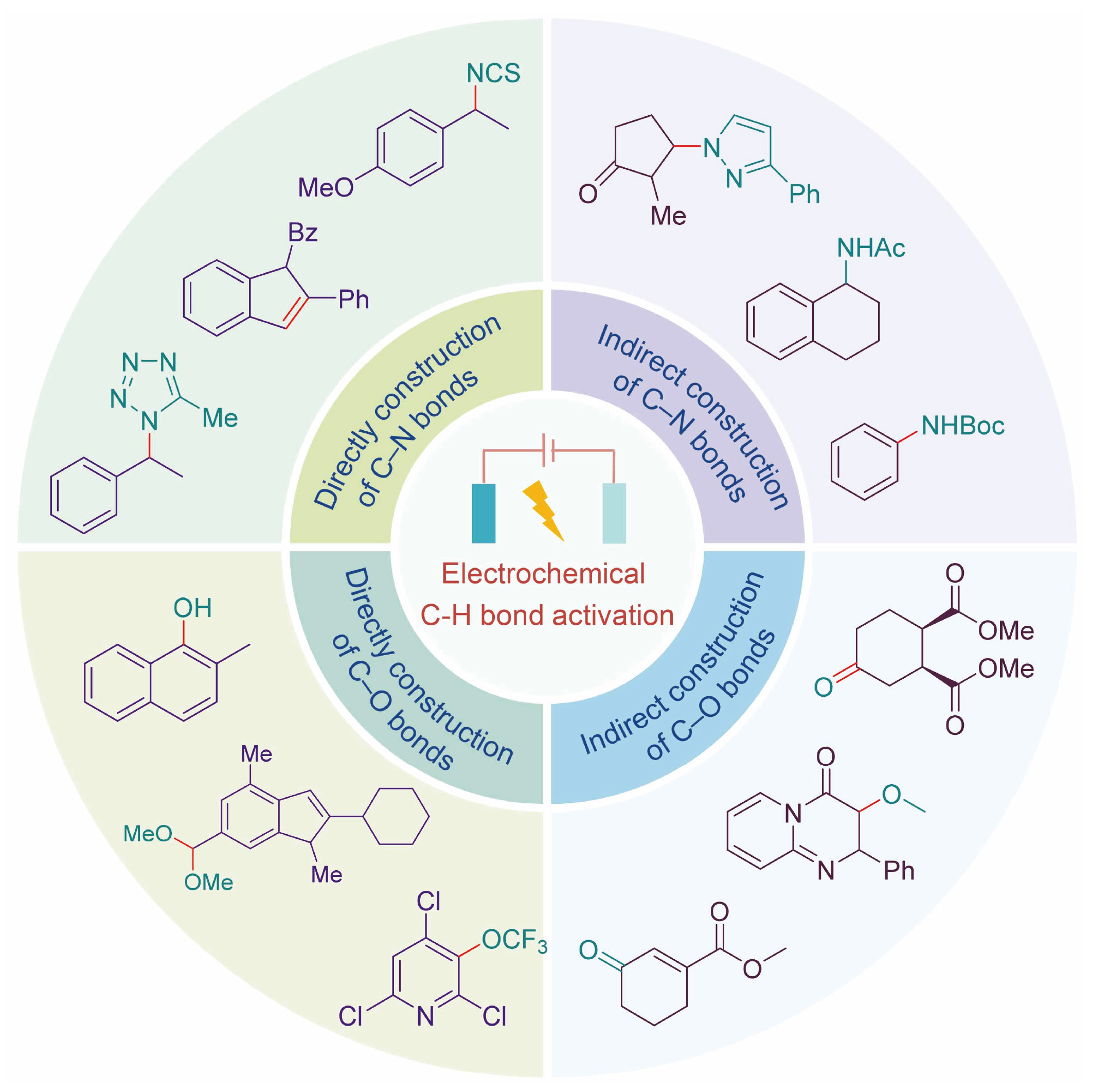
电化学C—H键活化以其优异的原子经济性、丰富的反应位点、环境友好性以及无需底物预官能团化等优势备受关注. C—N/C—O键广泛存在于许多天然产物和药物分子中, 是生命体系中重要的结构, 因此其高效构建方法的研究具有重要意义. 近年来, 随着绿色化学理念的深入, 无金属参与的电化学C—H键活化构建C—N/C—O键因其避免金属残留、成本低廉且环境友好等优势, 正日益成为研究热点. 该领域已开发出多种通过电化学C—H键活化构建C—N/C—O键策略. 系统总结了近五年来无金属电化学C—H键活化构建C—N/C—O键的重要进展, 涵盖直接C—N/C—O键的构建及非金属介导的C—N/C—O键的构建, 并重点阐述了相关反应机理.
罗辉, 王文权, 贺瑜, 王富强, 杨金会. 无金属电化学C—H键活化构建C—N/C—O键的研究进展[J]. 有机化学, 2026, 46(3): 697-724.
Hui Luo, Wenquan Wang, Yu He, Fuqiang Wang, Jinhui Yang. Research Progress in Metal-Free Electrochemical C—H Bond Activation for the Construction of C—N/C—O Bonds[J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 697-724.
| Item compared | Transition metal-catalyzed C—H functionalization | Metal-free electrochemical C—H functionalization |
|---|---|---|
| Reaction mechanism | Using transition metals as catalysts, through elementary steps such as ligand exchange, oxidative addition, migration insertion, and reductive elimination, the activation and functionalization of C—H bonds are achieved. | Using electrons as clean oxidants or reductants, through electron transfer at the electrode surface, the substrate is transformed into active intermediates, thereby achieving the cleavage and functionalization of C—H bonds. |
| Reactive driving force | Chemical oxidants (such as Ag salts, Cu salts, peroxides, etc.) or reductants provide or consume electrons to complete the catalytic cycle. | Electric energy. By applying an external voltage or current, the redox potential of the reaction can be precisely controlled. |
| major advantage | (1) The method is mature and has a wide range of applications; (2) It offers strong selectivity and can be precisely controlled through ligand engineering; (3) The reaction conditions are relatively mild and facilitate large-scale production. | (1) Green and sustainable, reducing the use of metals and oxidants; (2) Economical in steps, without the need for pre-functionalization steps; (3) Unique reactivity, facilitating the generation of highly reactive free radical species; (4) High safety, capable of avoiding some dangerous chemical oxidants. |
| Main challenges and limitations | (1) The cost of metals is high and they may be toxic; (2) Metal residues affect the purity of the products, especially in the pharmaceutical industry; (3) They rely on chemical reagents for oxidation, generating chemical waste. | (1) Selective control (especially regional and stereoscopic selectivity) is a major challenge; (2) Reaction scaling is difficult due to limitations imposed by electrode area and mass transfer efficiency; (3) Special equipment (electrolytic cells, power supplies) is required; (4) Requirements exist for the conductivity of the substrate and solvent. |
| Catalyst and cost | (1) Expensive transition metal catalysts (such as Pd, Rh, Ir) are required; (2) It may involve the design and synthesis of complex ligands; (3) There is a problem of metal residue, which is not friendly to drug synthesis. | (1) No need for transition metal catalysts, and the cost is relatively low; (2) It may use inexpensive media (such as salts, acids, and bases) to facilitate conductivity; (3) No metal residues, especially suitable for the fields of medicine and materials. |
| Reaction selecti- vity | Regional selectivity: Usually precisely controlled through the spatial/electronic effects of directing groups or ligands, as well as catalysts. Stereoselectivity: High enantioselectivity can be achie- ved through chiral ligands. | Regional selectivity: It can be regulated by substrate structure (such as inherent electronic effects at the site), electrode potential, and medium, but precise activation of the distal C—H bond poses greater challenges. Stereoselectivity: It is relatively difficult to control and is currently a research difficulty. |
| Sustainability and “Green Chemis- try” | Using stoichiometric oxidants results in poorer atom economy. (1) Toxic or expensive metals may be used; (2) The process has high economic efficiency, but the environmental footprint is relatively large. | The atomic economy is high, and electrons act as “clean reagents”. (1) It is more in line with the principles of green chemistry (reducing waste and using renewable sources); (2) It can be combined with renewable energy sources (solar energy, wind energy). |
| Item compared | Transition metal-catalyzed C—H functionalization | Metal-free electrochemical C—H functionalization |
|---|---|---|
| Reaction mechanism | Using transition metals as catalysts, through elementary steps such as ligand exchange, oxidative addition, migration insertion, and reductive elimination, the activation and functionalization of C—H bonds are achieved. | Using electrons as clean oxidants or reductants, through electron transfer at the electrode surface, the substrate is transformed into active intermediates, thereby achieving the cleavage and functionalization of C—H bonds. |
| Reactive driving force | Chemical oxidants (such as Ag salts, Cu salts, peroxides, etc.) or reductants provide or consume electrons to complete the catalytic cycle. | Electric energy. By applying an external voltage or current, the redox potential of the reaction can be precisely controlled. |
| major advantage | (1) The method is mature and has a wide range of applications; (2) It offers strong selectivity and can be precisely controlled through ligand engineering; (3) The reaction conditions are relatively mild and facilitate large-scale production. | (1) Green and sustainable, reducing the use of metals and oxidants; (2) Economical in steps, without the need for pre-functionalization steps; (3) Unique reactivity, facilitating the generation of highly reactive free radical species; (4) High safety, capable of avoiding some dangerous chemical oxidants. |
| Main challenges and limitations | (1) The cost of metals is high and they may be toxic; (2) Metal residues affect the purity of the products, especially in the pharmaceutical industry; (3) They rely on chemical reagents for oxidation, generating chemical waste. | (1) Selective control (especially regional and stereoscopic selectivity) is a major challenge; (2) Reaction scaling is difficult due to limitations imposed by electrode area and mass transfer efficiency; (3) Special equipment (electrolytic cells, power supplies) is required; (4) Requirements exist for the conductivity of the substrate and solvent. |
| Catalyst and cost | (1) Expensive transition metal catalysts (such as Pd, Rh, Ir) are required; (2) It may involve the design and synthesis of complex ligands; (3) There is a problem of metal residue, which is not friendly to drug synthesis. | (1) No need for transition metal catalysts, and the cost is relatively low; (2) It may use inexpensive media (such as salts, acids, and bases) to facilitate conductivity; (3) No metal residues, especially suitable for the fields of medicine and materials. |
| Reaction selecti- vity | Regional selectivity: Usually precisely controlled through the spatial/electronic effects of directing groups or ligands, as well as catalysts. Stereoselectivity: High enantioselectivity can be achie- ved through chiral ligands. | Regional selectivity: It can be regulated by substrate structure (such as inherent electronic effects at the site), electrode potential, and medium, but precise activation of the distal C—H bond poses greater challenges. Stereoselectivity: It is relatively difficult to control and is currently a research difficulty. |
| Sustainability and “Green Chemis- try” | Using stoichiometric oxidants results in poorer atom economy. (1) Toxic or expensive metals may be used; (2) The process has high economic efficiency, but the environmental footprint is relatively large. | The atomic economy is high, and electrons act as “clean reagents”. (1) It is more in line with the principles of green chemistry (reducing waste and using renewable sources); (2) It can be combined with renewable energy sources (solar energy, wind energy). |
| [1] |
doi: 10.1021/acscatal.3c02282 |
| [2] |
doi: 10.1002/cssc.202002397 pmid: 33207076 |
| [3] |
doi: 10.1021/acs.accounts.7b00306 |
| [4] |
doi: 10.1021/acs.chemrev.3c00207 |
| [5] |
doi: 10.1038/s41586-023-06347-3 |
| [6] |
doi: 10.1039/c5cs00628g pmid: 26507237 |
| [7] |
doi: 10.1021/ja407388y pmid: 24020940 |
| [8] |
doi: 10.1038/417507a |
| [9] |
doi: 10.1016/j.chempr.2021.12.001 |
| [10] |
doi: 10.1021/jacs.8b05195 |
| [11] |
doi: 10.1021/acs.accounts.3c00580 |
| [12] |
|
| [13] |
doi: 10.1002/cssc.v18.8 |
| [14] |
doi: 10.1039/D1CC06285A |
| [15] |
doi: 10.1021/acs.accounts.1c00547 |
| [16] |
doi: 10.1039/D1CS00923K |
| [17] |
doi: 10.1021/jacs.2c12151 |
| [18] |
|
| [19] |
doi: 10.1021/acscatal.3c05848 |
| [20] |
doi: 10.1021/acs.accounts.4c00656 |
| [21] |
doi: 10.1002/adsc.v363.7 |
| [22] |
doi: 10.1039/D1CS00150G |
| [23] |
doi: 10.1038/s41929-022-00855-7 |
| [24] |
doi: 10.1007/s11426-023-1735-x |
| [25] |
doi: 10.1021/acs.chemrev.3c00158 |
| [26] |
doi: 10.1002/cssc.v15.6 |
| [27] |
doi: 10.1039/d1cs00223f pmid: 34060564 |
| [28] |
doi: 10.1002/anie.v62.4 |
| [29] |
doi: 10.1021/acs.chemrev.8b00233 |
| [30] |
doi: 10.1021/acs.joc.1c00988 |
| [31] |
doi: 10.1039/D0CS00488J |
| [32] |
doi: 10.1002/cssc.v15.7 |
| [33] |
doi: 10.1039/d1cs00380a pmid: 34382989 |
| [34] |
doi: 10.1039/D1GC02922C |
| [35] |
doi: 10.1002/anie.v64.21 |
| [36] |
doi: 10.6023/cjoc202309024 |
|
(高瑞林, 文丽荣, 郭维斯, 有机化学, 2024, 44, 892.)
doi: 10.6023/cjoc202309024 |
|
| [37] |
doi: 10.1038/s41467-024-49223-y |
| [38] |
doi: 10.1021/jacs.3c08839 |
| [39] |
doi: 10.1002/anie.v63.3 |
| [40] |
doi: 10.1038/s41586-023-05886-z |
| [41] |
doi: 10.1039/D4CS01142B |
| [42] |
doi: 10.1002/anie.v59.29 |
| [43] |
doi: 10.1007/s10008-023-05507-9 |
| [44] |
doi: 10.1002/cssc.202000032 pmid: 32100961 |
| [45] |
doi: 10.1021/acssuschemeng.2c06046 |
| [46] |
doi: 10.1002/chem.v21.42 |
| [47] |
doi: 10.1016/j.chempr.2020.08.025 |
| [48] |
doi: 10.1002/adsu.v9.4 |
| [49] |
doi: 10.1002/anie.v57.16 |
| [50] |
doi: 10.1002/anie.v57.20 |
| [51] |
doi: 10.31635/ccschem.021.202101451 |
| [52] |
doi: 10.1039/D0SC01848A |
| [53] |
doi: 10.1021/acs.chemrev.9b00045 |
| [54] |
doi: 10.1039/D3CC01661G |
| [55] |
doi: 10.1021/acs.chemrev.9b00717 |
| [56] |
doi: 10.1002/advs.v12.28 |
| [57] |
doi: 10.1021/acs.joc.1c00931 |
| [58] |
doi: 10.1039/D0OB01008A |
| [59] |
doi: 10.1039/D3QO00280B |
| [60] |
doi: 10.6023/cjoc202310033 |
|
(朱子乐, 李鹏飞, 仇友爱, 有机化学, 2024, 44, 871.)
doi: 10.6023/cjoc202310033 |
|
| [61] |
doi: 10.1021/acs.orglett.2c00415 |
| [62] |
doi: 10.1002/ejoc.v2022.44 |
| [63] |
doi: 10.1002/anie.v60.6 |
| [64] |
doi: 10.1126/science.abf2798 pmid: 33542135 |
| [65] |
doi: 10.1039/D2SC05423J |
| [66] |
doi: 10.1021/acs.joc.3c00609 |
| [67] |
doi: 10.1055/a-1992-7066 |
| [68] |
doi: 10.1021/acs.orglett.3c04012 |
| [69] |
doi: 10.1021/acs.orglett.2c01782 |
| [70] |
doi: 10.1039/C8CC09899A |
| [71] |
doi: 10.1021/acs.joc.1c01842 |
| [72] |
doi: 10.1002/cssc.v15.23 |
| [73] |
doi: 10.1021/acs.orglett.3c02099 |
| [74] |
doi: 10.1039/D0GC00687D |
| [75] |
doi: 10.1039/D1GC04676D |
| [76] |
|
| [77] |
doi: 10.1021/acs.joc.4c00425 |
| [78] |
doi: 10.1021/acs.orglett.4c01463 |
| [79] |
doi: 10.1039/D1QO00746G |
| [80] |
doi: 10.1039/D3GC02126B |
| [81] |
doi: 10.1038/s41467-022-31813-3 pmid: 35842447 |
| [82] |
doi: 10.1039/D1CC05865G |
| [83] |
doi: 10.1021/jacs.3c11506 pmid: 38295054 |
| [84] |
doi: 10.1021/acs.joc.4c00502 |
| [85] |
doi: 10.1039/D0GC02294B |
| [86] |
doi: 10.1021/acs.joc.1c02947 |
| [87] |
doi: 10.1021/acs.joc.2c02363 |
| [88] |
doi: 10.1039/D0GC01871F |
| [89] |
|
| [90] |
doi: 10.1021/jacs.5c08109 |
| [91] |
|
| [92] |
doi: 10.1038/s41467-022-31634-4 pmid: 35803941 |
| [93] |
|
| [94] |
doi: 10.1002/anie.v62.40 |
| [95] |
doi: 10.1039/D4QO02022G |
| [96] |
doi: 10.1126/sciadv.aaz0590 |
| [97] |
doi: 10.1038/s41467-020-16519-8 pmid: 32483217 |
| [98] |
doi: 10.1021/jacs.1c00288 |
| [99] |
doi: 10.1002/celc.v8.20 |
| [100] |
doi: 10.1021/acs.orglett.1c02032 |
| [101] |
doi: 10.1002/ajoc.v10.11 |
| [102] |
doi: 10.1002/ejoc.v2020.11 |
| [103] |
|
| [104] |
doi: 10.1002/anie.v61.3 |
| [105] |
doi: 10.1039/D2CC03883H |
| [106] |
doi: 10.1021/acs.joc.1c02616 |
| [107] |
doi: 10.1021/acs.orglett.2c01930 |
| [108] |
doi: 10.1021/acs.joc.4c01632 |
| [109] |
doi: 10.1002/anie.v64.29 |
| [110] |
doi: 10.1038/s41586-022-05608-x |
| [111] |
doi: 10.1021/jacs.7b03539 pmid: 28510449 |
| [112] |
doi: 10.1002/ange.v130.22 |
| [113] |
doi: 10.1002/cctc.v16.22 |
| [114] |
doi: 10.1021/acs.joc.4c02793 |
| [1] | 马超, 徐晓玲, 骆翔, 鲁瑞娟, 虞国棋, 蔡涛. 电化学促进邻炔基苯甲酸酯与硫酚的自由基串联环化反应合成4-硫代异香豆素[J]. 有机化学, 2026, 46(3): 1039-1049. |
| [2] | 王馨玉, 黄晨佩, 郭昌, 宋琎. 电化学驱动芳基膦氧化物自由基二烯基化反应[J]. 有机化学, 2026, 46(3): 1050-1059. |
| [3] | 郭艳辉, 屈红恩, 梁佩. 手性有机硒化合物的合成研究进展[J]. 有机化学, 2026, 46(3): 786-805. |
| [4] | 曾依玲, 梁方鹏, 李辉, 刘荣荣, 李世清. α-氧代羧酸、炔和伯胺的多组分碳氢环化构筑异喹啉鎓骨架[J]. 有机化学, 2026, 46(2): 554-563. |
| [5] | 李静, 高峰, 张万斌. 抗肿瘤药物CDK4/6抑制剂合成工艺研究进展[J]. 有机化学, 2026, 46(2): 315-355. |
| [6] | 刘荣, 左应林, 张英俊, 张霁. “精准化学”在药物发现中的应用★[J]. 有机化学, 2025, 45(9): 3075-3097. |
| [7] | 张连吉, 王炳福, 潘大龙, 王翠苹, 赵玉辉, 迟海军, 董岩, 魏万国, 张志强. 乙酸介导的咪唑并吡啶与乙二醛水合物的无金属氧化C—H交叉偶联反应[J]. 有机化学, 2025, 45(8): 2923-2931. |
| [8] | 陈明, 张敬. 芳基羧酸无痕导向官能化反应的研究进展[J]. 有机化学, 2025, 45(8): 2660-2676. |
| [9] | 姚嫣, 付年凯. 光电化学金属催化研究进展[J]. 有机化学, 2025, 45(6): 1819-1837. |
| [10] | 周强, 杨宝臻, 郝贵林, 罗木鹏, 曹石, 赵蓓, 袁华, 王守国. 铑(III)催化的非活化烯烃与α-重氮羰基化合物的对映选择性烯丙位C—H键烷基化反应[J]. 有机化学, 2025, 45(6): 2109-2120. |
| [11] | 张艮红, 余若曦, 陈跃刚. 光/电促进醇及其衍生物C—O键活化构筑C(sp2)—C(sp3)键研究进展[J]. 有机化学, 2025, 45(5): 1548-1568. |
| [12] | 尤晓琴, 黄克金, 庄诗怡, 刘晨江, 金伟伟. 电化学促进的甘氨酸衍生物酯交换反应[J]. 有机化学, 2025, 45(4): 1360-1368. |
| [13] | 张朝威, 徐兵斌, 刘文龙, 赵敬, 段伟良. 钯催化不对称碳氢键活化合成平面手性二茂铁磺酰胺化合物[J]. 有机化学, 2025, 45(2): 707-716. |
| [14] | 刘泽水, 郭桢桢, 牛俊龙. 过渡金属催化C—H键硅基化反应构建硅杂环研究进展[J]. 有机化学, 2025, 45(2): 423-447. |
| [15] | 杨俊峰, 赵艳秋, 时磊. 电子供体-受体(EDA)复合物驱动的N-α位C—H键活化[J]. 有机化学, 2025, 45(2): 559-573. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||