有机化学 ›› 2026, Vol. 46 ›› Issue (3): 725-742.DOI: 10.6023/cjoc202509026 上一篇 下一篇
综述与进展
王雪倩a,b, 段宝忠b,*( ), 田红畅a,*(
), 田红畅a,*( ), 张磊a,*(
), 张磊a,*( )
)
收稿日期:2025-09-22
修回日期:2025-10-24
发布日期:2025-12-09
基金资助:
Xueqian Wanga,b, Baozhong Duanb,*( ), Hongchang Tiana,*(
), Hongchang Tiana,*( ), Lei Zhanga,*(
), Lei Zhanga,*( )
)
Received:2025-09-22
Revised:2025-10-24
Published:2025-12-09
Contact:
*Supported by:文章分享

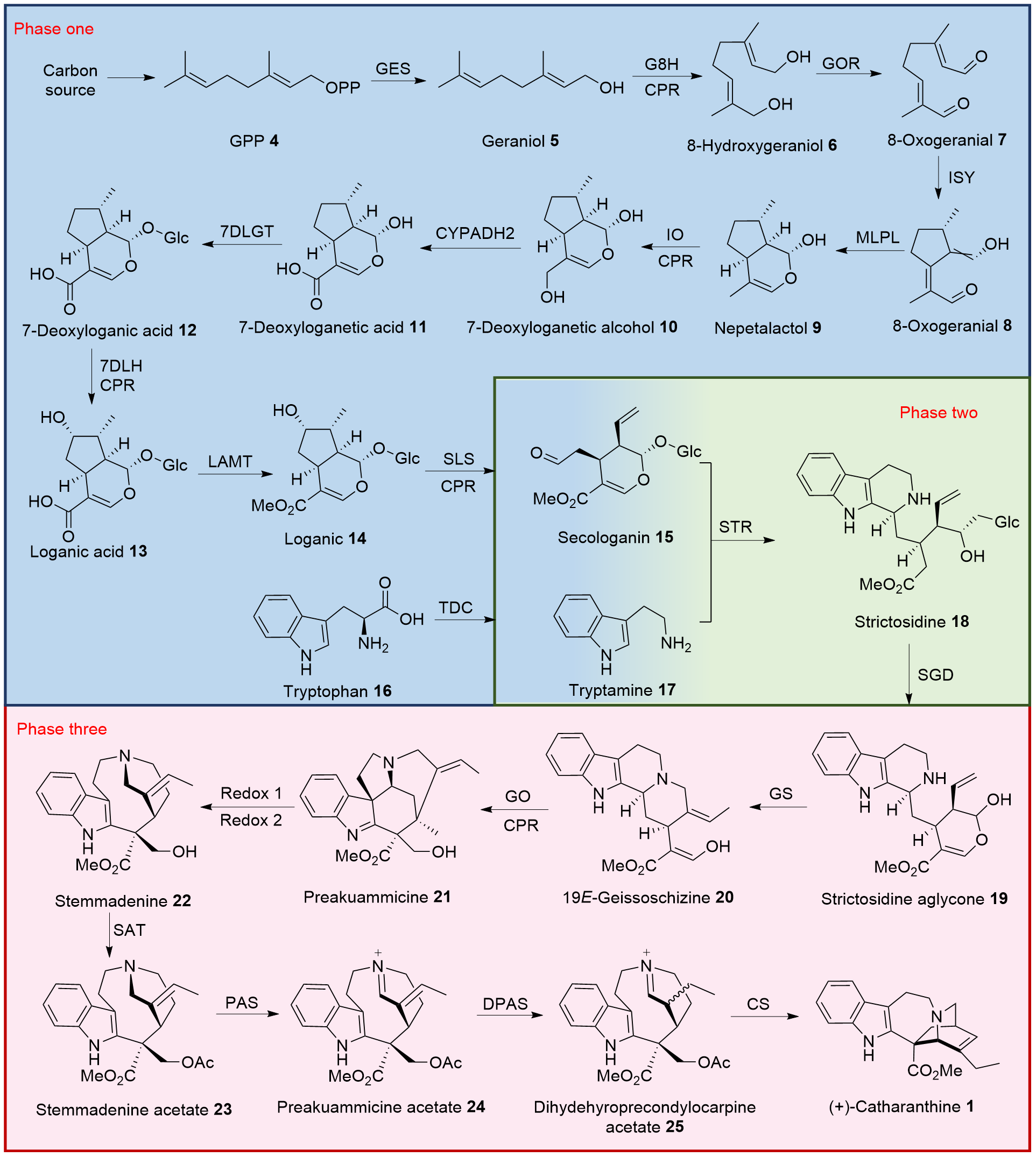
长春质碱(Catharanthine)是从长春花中分离得到的Iboga型吲哚生物碱, 具有五环结构, 三个手性中心, 是抗癌药物长春碱及其类似物长春新碱的重要结构片段, 因其复杂独特的化学结构成为合成研究的热点. 近年来, 其完整的生物合成途径已被解析, 并在酵母和本氏烟草等异源系统中实现了功能性重建. 自1969年以来, 已有18个研究团队报道了Catharanthine的化学合成路线, 涵盖消旋合成、不对称合成、仿生合成及多种过渡金属催化方法. 按照时间顺序综述了Catharanthine的生物合成与化学合成研究进展, 并对其未来发展方向进行了展望.
王雪倩, 段宝忠, 田红畅, 张磊. 吲哚生物碱Catharanthine合成研究进展[J]. 有机化学, 2026, 46(3): 725-742.
Xueqian Wang, Baozhong Duan, Hongchang Tian, Lei Zhang. Research Progress in Synthesis of Indole Alkaloid Catharanthine[J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 725-742.
| Entry | Research group | Year | (±/+/-)-Catharanthine | Steps | Yield/% | Reference |
|---|---|---|---|---|---|---|
| 1 | Büchiʼ group | 1969 | (±)-Catharanthine | 17 | 1.4 | [ |
| 2 | Kutneyʼ group | 1975 | (±)-Catharanthine | 12 | <1 | [ |
| 3 | Trostʼ group | 1979 | (±)-Catharanthine | 12 | 13 | [ |
| 4 | Beislerʼ group | 1980 | (±)-Catharanthine | 14 | <1 | [ |
| 5 | Tmanishiʼ group | 1980 | (±)-Catharanthine | 19 | <1 | [ |
| 6 | Dasʼ group | 1981 | (±)-Catharanthine | 10 | 1.4 | [ |
| 7 | Raucherʼ group | 1985 | (±)-Catharanthine | 10 | 13 | [ |
| 8 | Kuehneʼ group | 1986 | (±)-Catharanthine | 9 | 11 | [ |
| 9 | Kuehneʼ group | 2008 | (±)-Catharanthine | 12 | 2.6 | [ |
| 10 | Szántayʼ group | 1990 | (+)-Catharanthine | 9 | 6.3 | [ |
| 11 | Fukuyamaʼ group | 1999 | (±)-Catharanthine | 13 | 12.4 | [ |
| 12 | Dorisʼ group | 2006 | (+)-Catharanthine | 24 | <1 | [ |
| 13 | Oguriʼ group | 2013 | (-)-Catharanthine | 10 | 3.2 | [ |
| 14 | Ishiharaʼ group | 2014 | (-)-Catharanthine | 17 | <1 | [ |
| 15 | Ishiharaʼ group | 2015 | (+)-Catharanthine | 19 | <1 | [ |
| 16 | Tuoping Luoʼ group | 2016 | (+)-Catharanthine | 13 | <1 | [ |
| 17 | Bateyʼ group | 2018 | (+)-Catharanthine | 15 | <1 | [ |
| 18 | Nemotoʼ group | 2019 | (+)-Catharanthine | 27 | <1 | [ |
| 19 | Dixonʼ group | 2021 | (±)-Catharanthine | 5 | 2.1 | [ |
| 20 | Grögerʼ group | 2023 | (-)-Catharanthine | 22 | <1 | [ |
| Entry | Research group | Year | (±/+/-)-Catharanthine | Steps | Yield/% | Reference |
|---|---|---|---|---|---|---|
| 1 | Büchiʼ group | 1969 | (±)-Catharanthine | 17 | 1.4 | [ |
| 2 | Kutneyʼ group | 1975 | (±)-Catharanthine | 12 | <1 | [ |
| 3 | Trostʼ group | 1979 | (±)-Catharanthine | 12 | 13 | [ |
| 4 | Beislerʼ group | 1980 | (±)-Catharanthine | 14 | <1 | [ |
| 5 | Tmanishiʼ group | 1980 | (±)-Catharanthine | 19 | <1 | [ |
| 6 | Dasʼ group | 1981 | (±)-Catharanthine | 10 | 1.4 | [ |
| 7 | Raucherʼ group | 1985 | (±)-Catharanthine | 10 | 13 | [ |
| 8 | Kuehneʼ group | 1986 | (±)-Catharanthine | 9 | 11 | [ |
| 9 | Kuehneʼ group | 2008 | (±)-Catharanthine | 12 | 2.6 | [ |
| 10 | Szántayʼ group | 1990 | (+)-Catharanthine | 9 | 6.3 | [ |
| 11 | Fukuyamaʼ group | 1999 | (±)-Catharanthine | 13 | 12.4 | [ |
| 12 | Dorisʼ group | 2006 | (+)-Catharanthine | 24 | <1 | [ |
| 13 | Oguriʼ group | 2013 | (-)-Catharanthine | 10 | 3.2 | [ |
| 14 | Ishiharaʼ group | 2014 | (-)-Catharanthine | 17 | <1 | [ |
| 15 | Ishiharaʼ group | 2015 | (+)-Catharanthine | 19 | <1 | [ |
| 16 | Tuoping Luoʼ group | 2016 | (+)-Catharanthine | 13 | <1 | [ |
| 17 | Bateyʼ group | 2018 | (+)-Catharanthine | 15 | <1 | [ |
| 18 | Nemotoʼ group | 2019 | (+)-Catharanthine | 27 | <1 | [ |
| 19 | Dixonʼ group | 2021 | (±)-Catharanthine | 5 | 2.1 | [ |
| 20 | Grögerʼ group | 2023 | (-)-Catharanthine | 22 | <1 | [ |
| [1] |
doi: 10.1073/pnas.2318586121 |
| [2] |
doi: 10.1124/jpet.112.199661 pmid: 23532933 |
| [3] |
doi: 10.1021/acschemneuro.3c00478 |
| [4] |
doi: 10.1007/s00210-024-03191-8 |
| [5] |
doi: 10.1002/jps.3030480419 |
| [6] |
(a)
doi: 10.1021/acs.orglett.7b03694 |
|
(b)
doi: 10.1038/s41586-022-05157-3 |
|
| [7] |
(a)
doi: 10.1007/s00253-020-10592-1 pmid: 15032608 |
|
(b)
pmid: 15032608 |
|
|
(c)
doi: 10.2174/0929867043455846 pmid: 15032608 |
|
| [8] |
(a)
pmid: 18609590 |
|
(b)
doi: 10.1021/ar50029a002 pmid: 18609590 |
|
|
(c)
pmid: 18609590 |
|
|
(d)
doi: 10.1007/BF00233027 pmid: 18609590 |
|
|
(e)
pmid: 18609590 |
|
|
(f)
doi: 10.1016/S0922-338X(97)80984-1 pmid: 18609590 |
|
|
(g)
doi: 10.1016/S0141-0229(01)00306-4 pmid: 18609590 |
|
|
(h)
doi: 10.1007/s00299-010-0874-0 pmid: 18609590 |
|
| [9] |
doi: 10.1126/science.aat4100 |
| [10] |
doi: 10.1021/ol200996a |
| [11] |
doi: 10.4014/jmb.2207.07057 |
| [12] |
(a)
doi: 10.1073/pnas.1501821112 pmid: 34302444 |
|
(b)
doi: 10.1002/bit.v119.5 pmid: 34302444 |
|
|
(c)
doi: 10.1111/1751-7915.13898 pmid: 34302444 |
|
| [13] |
doi: 10.1038/s44160-022-00205-2 |
| [14] |
(a)
doi: 10.1146/phyto.2018.56.issue-1 |
|
(b)
doi: 10.1007/s00299-018-2296-3 |
|
| [15] |
doi: 10.1016/j.molp.2023.10.016 |
| [16] |
doi: 10.1093/mp/ssq020 pmid: 20457641 |
| [17] |
doi: 10.1038/s41586-020-2546-8 |
| [18] |
doi: 10.1021/acssynbio.2c00434 |
| [19] |
doi: 10.1021/ja00707a043 |
| [20] |
pmid: 5459202 |
| [21] |
(a)
doi: 10.1021/ja01065a038 pmid: 5418451 |
|
(b)
doi: 10.1021/ja00972a061 pmid: 5418451 |
|
|
(c)
pmid: 5418451 |
|
|
(d)
pmid: 5418451 |
|
| [22] |
doi: 10.1021/jo01326a044 |
| [23] |
doi: 10.1016/0040-4020(80)80061-5 |
| [24] |
pmid: 6037875 |
| [25] |
doi: 10.1021/ja01059a063 |
| [26] |
|
| [27] |
|
| [28] |
pmid: 1176299 |
| [29] |
doi: 10.1021/jo00217a052 |
| [30] |
(a)
doi: 10.1021/jo00365a012 |
|
(b)
doi: 10.1016/j.tet.2008.07.044 |
|
| [31] |
doi: 10.1016/S0040-4020(01)81977-3 |
| [32] |
(a)
doi: 10.1021/ol990749i |
|
(b)
doi: 10.3987/COM-01-S(K)42 |
|
| [33] |
doi: 10.1002/anie.v45:32 |
| [34] |
doi: 10.1038/nchem.1798 pmid: 24345948 |
| [35] |
doi: 10.1039/C4CC01445F |
| [36] |
doi: 10.1021/jacs.5b08693 |
| [37] |
doi: 10.1039/c6sc00932h pmid: 30034694 |
| [38] |
pmid: 17941641 |
| [39] |
doi: 10.1039/C8QO00849C |
| [40] |
doi: 10.1021/acs.orglett.9b01198 |
| [41] |
doi: 10.1021/ja00236a023 |
| [42] |
doi: 10.1021/jacs.1c04980 pmid: 34254792 |
| [43] |
doi: 10.1055/a-2086-0690 |
| [1] | 张涛, 季珍瑜, 唐功利, 侯现锋. 金色链霉菌SP-371中一个新型倍半萜内酯化合物的发现[J]. 有机化学, 2026, 46(2): 689-692. |
| [2] | 程文富, 赵明豪, 杨宇晨, 王丽佳. N-Methyl Pratensilin B分子的首次全合成[J]. 有机化学, 2026, 46(2): 670-677. |
| [3] | 何卫刚, 段明月, 闫帅, 叶伟杰, 徐雪园, 孙先宇. 蕊木属生物碱arboridinine和arborisidine的全合成研究进展[J]. 有机化学, 2026, 46(2): 443-454. |
| [4] | 赵礼麟, 李兆麒, 钟李平, 闵龙. abeo-甾体的全合成研究进展[J]. 有机化学, 2026, 46(1): 21-38. |
| [5] | 崔心怡, 郭丽帆, 马聪璇, 李耘, 梁建华. 抗耐药菌红霉素的结构修饰策略、构效关系及全合成研究进展[J]. 有机化学, 2026, 46(1): 39-73. |
| [6] | 赵倩倩, 魏培垚, 刘孙典, 张博鑫, 梁承远. 钯催化串联反应在含季碳中心的复杂天然产物全合成中的应用[J]. 有机化学, 2025, 45(9): 3289-3300. |
| [7] | 夏颖, 朱辰龙, 孙炳峰. 抗乙肝病毒药物恩替卡韦的全合成研究进展★[J]. 有机化学, 2025, 45(9): 3186-3202. |
| [8] | 刘小宇, 许庭瑞, 秦勇. 吗啡类生物碱的全合成研究新进展★[J]. 有机化学, 2025, 45(9): 3098-3112. |
| [9] | 莫百川, 李婷婷, 王芳, 李晓杰, 巴达日夫. 咔唑醌天然产物的合成研究进展[J]. 有机化学, 2025, 45(8): 2746-2766. |
| [10] | 丁一鸣, 张庭荣, 张泾渭, 邓军. 三尖杉二萜类天然产物的全合成研究进展[J]. 有机化学, 2025, 45(6): 2048-2073. |
| [11] | 何卫刚, 刘亚东, 邓迎开, 孙先宇. Malagasy生物碱的合成研究进展[J]. 有机化学, 2025, 45(4): 1153-1165. |
| [12] | 易九州, 火亮, 陈金燕, 刘勐, 李辉林, 厍学功. 保护基效应对几类天然产物全合成的影响[J]. 有机化学, 2025, 45(3): 1030-1039. |
| [13] | 陈杰, 李俊, 龙先文, 申海香, 邓军. Wagner-Meerwein重排反应在天然产物全合成中的应用[J]. 有机化学, 2025, 45(3): 896-912. |
| [14] | 李闯, 张成, 刘小宇, 秦勇. 二萜生物碱全合成研究进展[J]. 有机化学, 2025, 45(3): 881-895. |
| [15] | 杨庆星, 刘璇, 马硕, 李欣欣, 马东旭, 徐涛. 多卤代海洋来源天然产物全合成[J]. 有机化学, 2025, 45(3): 764-803. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||