有机化学 ›› 2026, Vol. 46 ›› Issue (4): 1677-1684.DOI: 10.6023/cjoc202512046 上一篇 下一篇
研究论文
收稿日期:2025-12-29
修回日期:2026-03-14
发布日期:2026-04-02
通讯作者:
李斌栋, 王定海
基金资助:
Ziliang Yuan, Bingdong Li*( ), Dinghai Wang*(
), Dinghai Wang*( )
)
Received:2025-12-29
Revised:2026-03-14
Published:2026-04-02
Contact:
Bingdong Li, Dinghai Wang
Supported by:文章分享
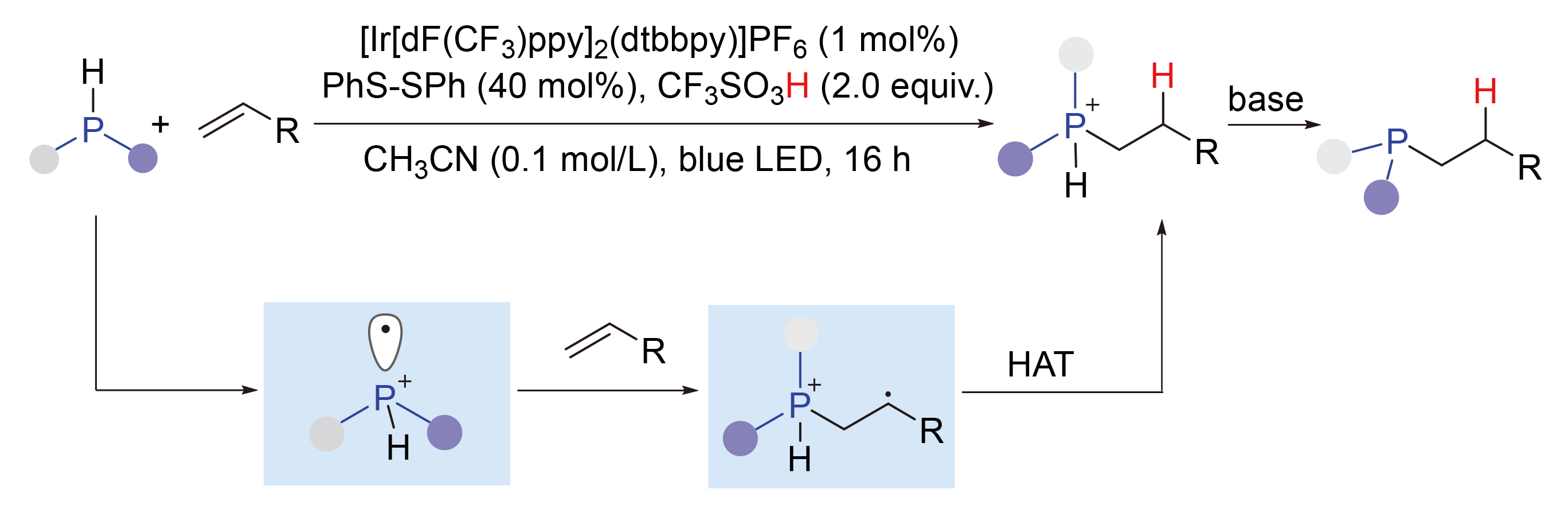
烯烃的氢膦化反应是有机合成中构建P—C键的核心策略. 发展了一种通过可见光催化烯烃的氢膦化合成叔膦化合物的新方法, 该反应显示出广泛的底物范围和优异的官能团相容性, 以中等至优异的收率(48%~95%)生成各种叔膦化合物. 提出了合理的反应机理: 光激发催化剂促使二苯基膦发生单电子氧化, 生成相应的膦自由基阳离子(PRC), 该中间体随即与烯烃的碳-碳双键发生自由基加成反应. 此外, 通过核磁共振(NMR)光谱成功监测到了季鏻盐和三价膦的生成, 为反应机理提供了直接佐证.
袁子亮, 李斌栋, 王定海. 可见光催化的烯烃氢膦化反应[J]. 有机化学, 2026, 46(4): 1677-1684.
Ziliang Yuan, Bingdong Li, Dinghai Wang. Visible-Light Induced Hydrophosphination of Olefins[J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1677-1684.
| Entry | Variation from the “standard conditions” | Yieldb/% |
|---|---|---|
| 1 | None | 89 |
| 2 | 2,4,6-Trimethylbenzenethiol in place of PhS-SPh | 31 |
| 3 | CF3SO3H (1.0 equiv.) was used | 60 |
| 4 | CH3COOH (1.0 equiv.) in place of CF3SO3H | 23 |
| 5 | CF3COOH (1.0 equiv.) in place of CF3SO3H | 26 |
| 6 | 2a (1.5 equiv.) was used | 39 |
| 7 | Ir(ppy)3 in place of [Ir[dF(CF3)ppy]2(dtbbpy)]PF6 | 22 |
| 8 | 4-CzIPN in place of [Ir[dF(CF3)ppy]2(dtbbpy)]PF6 | 12 |
| 9 | Eosin Y in place of [Ir[dF(CF3)ppy]2(dtbbpy)]PF6 | 3 |
| 10 | DCE in place of CH3CN | 49 |
| 11 | DMF in place of CH3CN | 41 |
| 12 | THF in place of CH3CN | 14 |
| 13 | Without [Ir[dF(CF3)ppy]2(dtbbpy)]PF6 | 5 |
| 14 | Without light | 0 |
| Entry | Variation from the “standard conditions” | Yieldb/% |
|---|---|---|
| 1 | None | 89 |
| 2 | 2,4,6-Trimethylbenzenethiol in place of PhS-SPh | 31 |
| 3 | CF3SO3H (1.0 equiv.) was used | 60 |
| 4 | CH3COOH (1.0 equiv.) in place of CF3SO3H | 23 |
| 5 | CF3COOH (1.0 equiv.) in place of CF3SO3H | 26 |
| 6 | 2a (1.5 equiv.) was used | 39 |
| 7 | Ir(ppy)3 in place of [Ir[dF(CF3)ppy]2(dtbbpy)]PF6 | 22 |
| 8 | 4-CzIPN in place of [Ir[dF(CF3)ppy]2(dtbbpy)]PF6 | 12 |
| 9 | Eosin Y in place of [Ir[dF(CF3)ppy]2(dtbbpy)]PF6 | 3 |
| 10 | DCE in place of CH3CN | 49 |
| 11 | DMF in place of CH3CN | 41 |
| 12 | THF in place of CH3CN | 14 |
| 13 | Without [Ir[dF(CF3)ppy]2(dtbbpy)]PF6 | 5 |
| 14 | Without light | 0 |
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [1] | 侯馨怡, 史同同, 李泽江, 董庆昊, 孙凯, 王薪. 可见光诱导的磷酰化环化合成磷酰基取代的吲哚并二氮䓬[J]. 有机化学, 2026, 46(4): 1776-1787. |
| [2] | 郑婧斐, 吕琪妍, 孙凯, 陈晓岚, 屈凌波, 王金泉, 於兵. 自由基介导的6-氮杂尿嘧啶官能团化研究进展[J]. 有机化学, 2026, 46(4): 1603-1620. |
| [3] | 赵雨晴, 朱先进, 石凌宇, 魏鸣宇, 徐紫涵, 石永佳, 李旭锋, 杨道山. 环硫鎓盐开环官能化反应的研究进展[J]. 有机化学, 2026, 46(4): 1572-1602. |
| [4] | 张皓然, 贾均松, 俞琼, 吴宇, 李玉龙, 舒伟. 可见光催化烯烃的氢胺化和氢(磺)酰胺化反应[J]. 有机化学, 2026, 46(4): 1166-1180. |
| [5] | 杨珊, 陈亚苏, 朱晨. 自由基介导的官能团迁移-环化构建含氮稠杂芳烃[J]. 有机化学, 2026, 46(4): 1739-1749. |
| [6] | 卢琳玉, 亓雨彤, 张庭珲, 金胜男, 曹中艳, 张洪伟. 自由基介导烯烃的1,2,n-三官能团化反应研究进展[J]. 有机化学, 2026, 46(4): 1540-1559. |
| [7] | 董建洋, 薛东. 自由基途径的[1.1.1]螺桨烷官能化研究进展[J]. 有机化学, 2026, 46(4): 1464-1480. |
| [8] | 孟令旭, 申霖, 罗祥玲, 林玉妹, 龚磊. 具有光活性的铬、钴配合物的设计及其在有机合成中的应用进展[J]. 有机化学, 2026, 46(4): 1513-1528. |
| [9] | 苏雯鑫, 崔浩, 张霄. 单质硫(S8)参与的光化学反应研究进展[J]. 有机化学, 2026, 46(4): 1360-1375. |
| [10] | 刘琼, 杨修龙, 李浩源, 沈佳慧, 杨硕, 孟庆元, 牛效莹. 盐酸-二甲基亚砜体系促进酰胺的需氧光催化N-脱甲基反应[J]. 有机化学, 2026, 46(4): 1713-1721. |
| [11] | 余宁, 周于强, 魏晔. 光催化自由基介导的内酯合成研究进展[J]. 有机化学, 2026, 46(4): 1222-1254. |
| [12] | 张斌, 肖文精, 陈加荣. 烯烃自由基阴离子的可控产生与转化[J]. 有机化学, 2026, 46(4): 1181-1204. |
| [13] | 杨其昌, 张筱睿, 吕剑, 古双喜. 糖羟基的选择性氧化反应研究进展[J]. 有机化学, 2026, 46(3): 743-758. |
| [14] | 郭艳辉, 屈红恩, 梁佩. 手性有机硒化合物的合成研究进展[J]. 有机化学, 2026, 46(3): 786-805. |
| [15] | 任钶, 张光露, 牛轶凡, 王晓萌, 陈灿玉, 蒋敏. 光催化合成含氮杂芳环非天然氨基酸[J]. 有机化学, 2026, 46(1): 215-224. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||