有机化学 ›› 2026, Vol. 46 ›› Issue (4): 1603-1620.DOI: 10.6023/cjoc202510017 上一篇 下一篇
综述与进展
郑婧斐a, 吕琪妍a,b,*( ), 孙凯a, 陈晓岚a, 屈凌波a,c, 王金泉d,*(
), 孙凯a, 陈晓岚a, 屈凌波a,c, 王金泉d,*( ), 於兵a,*(
), 於兵a,*( )
)
收稿日期:2025-10-23
修回日期:2025-12-05
发布日期:2026-01-23
通讯作者:
吕琪妍, 王金泉, 於兵
基金资助:
Jingfei Zhenga, Qiyan Lva,b,*( ), Kai Suna, Xiaolan Chena, Lingbo Qua,c, Jinquan Wangd,*(
), Kai Suna, Xiaolan Chena, Lingbo Qua,c, Jinquan Wangd,*( ), Bing Yua,*(
), Bing Yua,*( )
)
Received:2025-10-23
Revised:2025-12-05
Published:2026-01-23
Contact:
Qiyan Lv, Jinquan Wang, Bing Yu
Supported by:文章分享
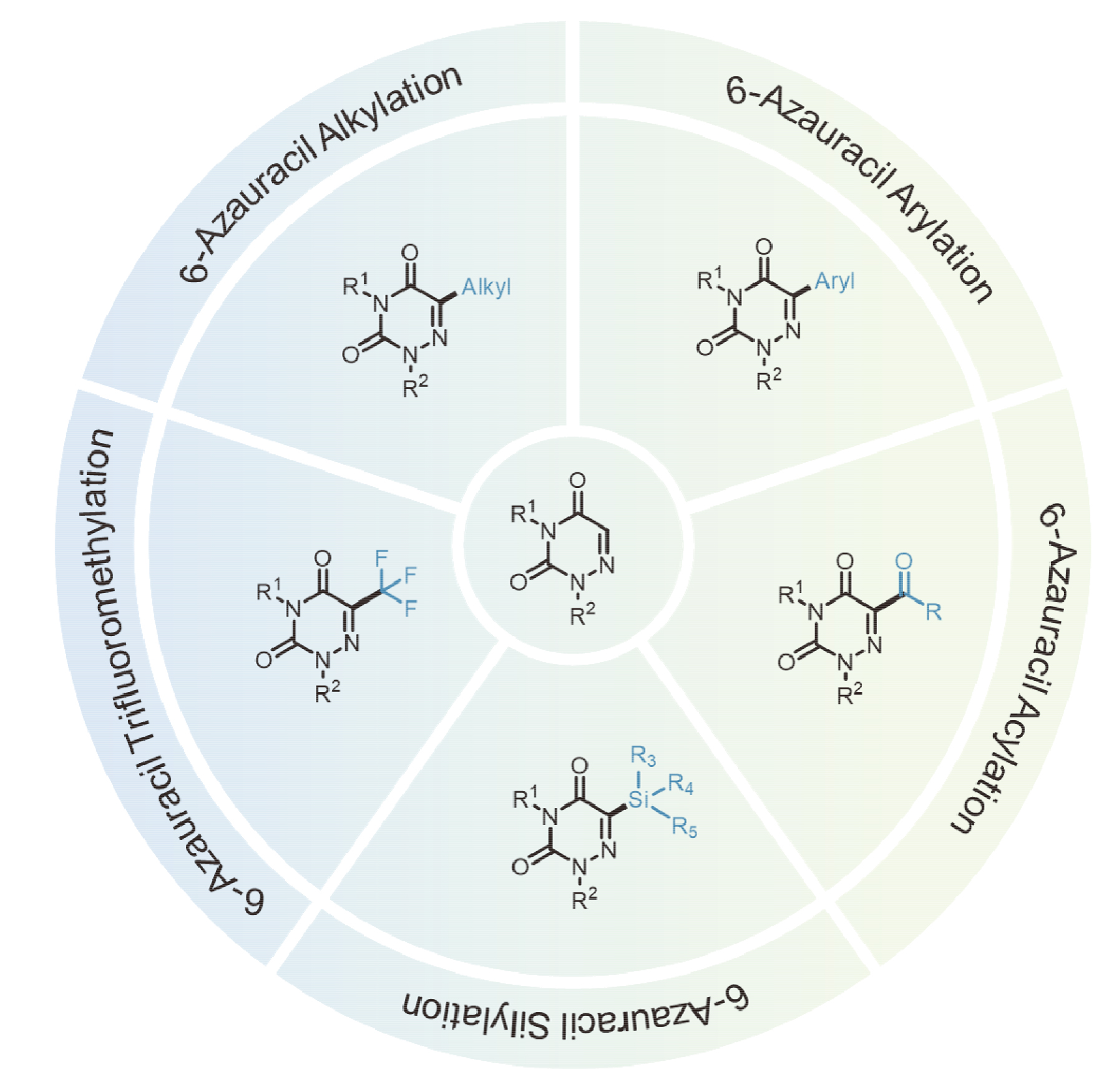
6-氮杂尿嘧啶作为尿嘧啶的关键类似物, 其骨架是多种生物活性分子的核心结构单元, 因此发展其结构修饰新方法具有重要意义. 近年来, 随着光催化和电催化等绿色、温和的自由基新策略的日益成熟, 6-氮杂尿嘧啶的自由基官能团化研究已取得重要进展. 此文聚焦于2021年以来6-氮杂尿嘧啶通过自由基反应实现的烷基化、芳基化、酰基化、硅基化及三氟甲基化等研究进展, 重点综述其新反应方法的设计与机理创新.
郑婧斐, 吕琪妍, 孙凯, 陈晓岚, 屈凌波, 王金泉, 於兵. 自由基介导的6-氮杂尿嘧啶官能团化研究进展[J]. 有机化学, 2026, 46(4): 1603-1620.
Jingfei Zheng, Qiyan Lv, Kai Sun, Xiaolan Chen, Lingbo Qu, Jinquan Wang, Bing Yu. Recent Advances in Functionalization of 6-Azauracils via Radical Reactions[J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1603-1620.
| [1] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
(a)
|
|
(b)
|
|
| [6] |
|
| [7] |
|
| [8] |
(a)
|
|
(方家恒, 田润妍, 陈继君, 刘心元, 有机化学, 2025, 45, 22.)
|
|
|
(b)
|
|
| [9] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [10] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(辛翠, 何卫民, 有机化学, 2024, 44, 3790.)
|
|
|
(f)
|
|
|
(g)
|
|
|
(h)
|
|
| [11] |
|
| [12] |
|
| [13] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [18] |
(a)
|
|
(b)
|
|
| [19] |
|
| [20] |
(a)
|
|
(b)
|
|
| [21] |
|
| [22] |
|
| [23] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [24] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(相盼洁, 於兵, 有机化学, 2024, 44, 2057.)
|
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
(a)
|
| [38] |
|
| [39] |
|
| [40] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [41] |
(a)
|
|
(b)
|
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(刘帅, 陈坤泉, 孙德群, 刘强, 陈祥雨, 有机化学, 2025, 45, 2545.)
|
|
|
(e)
|
|
|
(f)
|
|
| [49] |
|
| [50] |
|
| [1] | 侯馨怡, 史同同, 李泽江, 董庆昊, 孙凯, 王薪. 可见光诱导的磷酰化环化合成磷酰基取代的吲哚并二氮䓬[J]. 有机化学, 2026, 46(4): 1776-1787. |
| [2] | 王齐翔, 陈圣希, 王静, 陈栋, 鲍晓光, 吴新鑫. 无金属参与的多烯选择性氯代反应[J]. 有机化学, 2026, 46(4): 1750-1762. |
| [3] | 刘世峥, 窦明煜, 崔玉, 窦建民, 魏文廷. 自由基硒磺酰化研究进展[J]. 有机化学, 2026, 46(4): 1621-1634. |
| [4] | 赵雨晴, 朱先进, 石凌宇, 魏鸣宇, 徐紫涵, 石永佳, 李旭锋, 杨道山. 环硫鎓盐开环官能化反应的研究进展[J]. 有机化学, 2026, 46(4): 1572-1602. |
| [5] | 张皓然, 贾均松, 俞琼, 吴宇, 李玉龙, 舒伟. 可见光催化烯烃的氢胺化和氢(磺)酰胺化反应[J]. 有机化学, 2026, 46(4): 1166-1180. |
| [6] | 杨珊, 陈亚苏, 朱晨. 自由基介导的官能团迁移-环化构建含氮稠杂芳烃[J]. 有机化学, 2026, 46(4): 1739-1749. |
| [7] | 卢琳玉, 亓雨彤, 张庭珲, 金胜男, 曹中艳, 张洪伟. 自由基介导烯烃的1,2,n-三官能团化反应研究进展[J]. 有机化学, 2026, 46(4): 1540-1559. |
| [8] | 唐玉青, 杨紫娴, 杨兵, 王毅, 杜炳南. 球磨条件下机械力驱动的自由基反应研究进展[J]. 有机化学, 2026, 46(4): 1111-1145. |
| [9] | 董建洋, 薛东. 自由基途径的[1.1.1]螺桨烷官能化研究进展[J]. 有机化学, 2026, 46(4): 1464-1480. |
| [10] | 孟令旭, 申霖, 罗祥玲, 林玉妹, 龚磊. 具有光活性的铬、钴配合物的设计及其在有机合成中的应用进展[J]. 有机化学, 2026, 46(4): 1513-1528. |
| [11] | 苏雯鑫, 崔浩, 张霄. 单质硫(S8)参与的光化学反应研究进展[J]. 有机化学, 2026, 46(4): 1360-1375. |
| [12] | 刘琼, 杨修龙, 李浩源, 沈佳慧, 杨硕, 孟庆元, 牛效莹. 盐酸-二甲基亚砜体系促进酰胺的需氧光催化N-脱甲基反应[J]. 有机化学, 2026, 46(4): 1713-1721. |
| [13] | 吉伟文, 金健. 配体-金属电荷转移驱动的光促金属催化芳基羧酸脱羧转化[J]. 有机化学, 2026, 46(4): 1320-1329. |
| [14] | 余宁, 周于强, 魏晔. 光催化自由基介导的内酯合成研究进展[J]. 有机化学, 2026, 46(4): 1222-1254. |
| [15] | 袁子亮, 李斌栋, 王定海. 可见光催化的烯烃氢膦化反应[J]. 有机化学, 2026, 46(4): 1677-1684. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||