有机化学 ›› 2026, Vol. 46 ›› Issue (3): 1017-1026.DOI: 10.6023/cjoc202508002 上一篇 下一篇
研究论文
收稿日期:2025-08-03
修回日期:2025-10-05
发布日期:2025-11-27
基金资助:
Yu Sun, Yantao Zhu, Dong Zhang, Chunjie Ni*( )
)
Received:2025-08-03
Revised:2025-10-05
Published:2025-11-27
Contact:
*E-mail: Supported by:文章分享
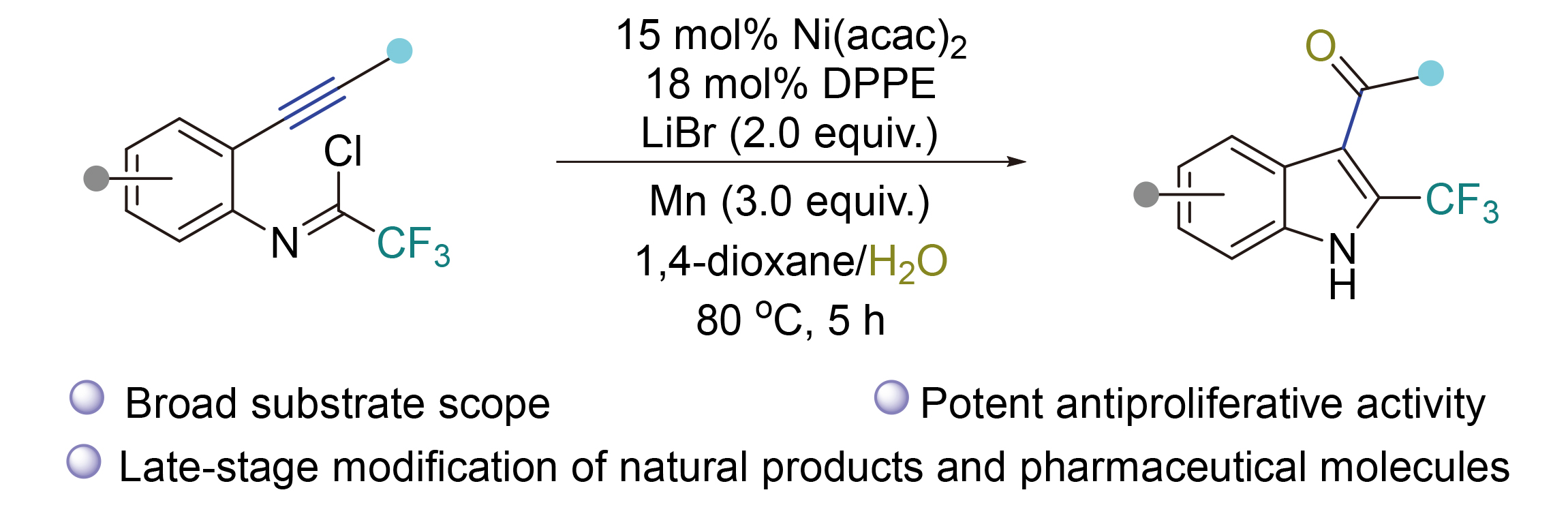
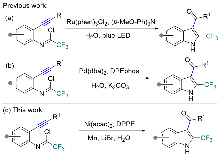
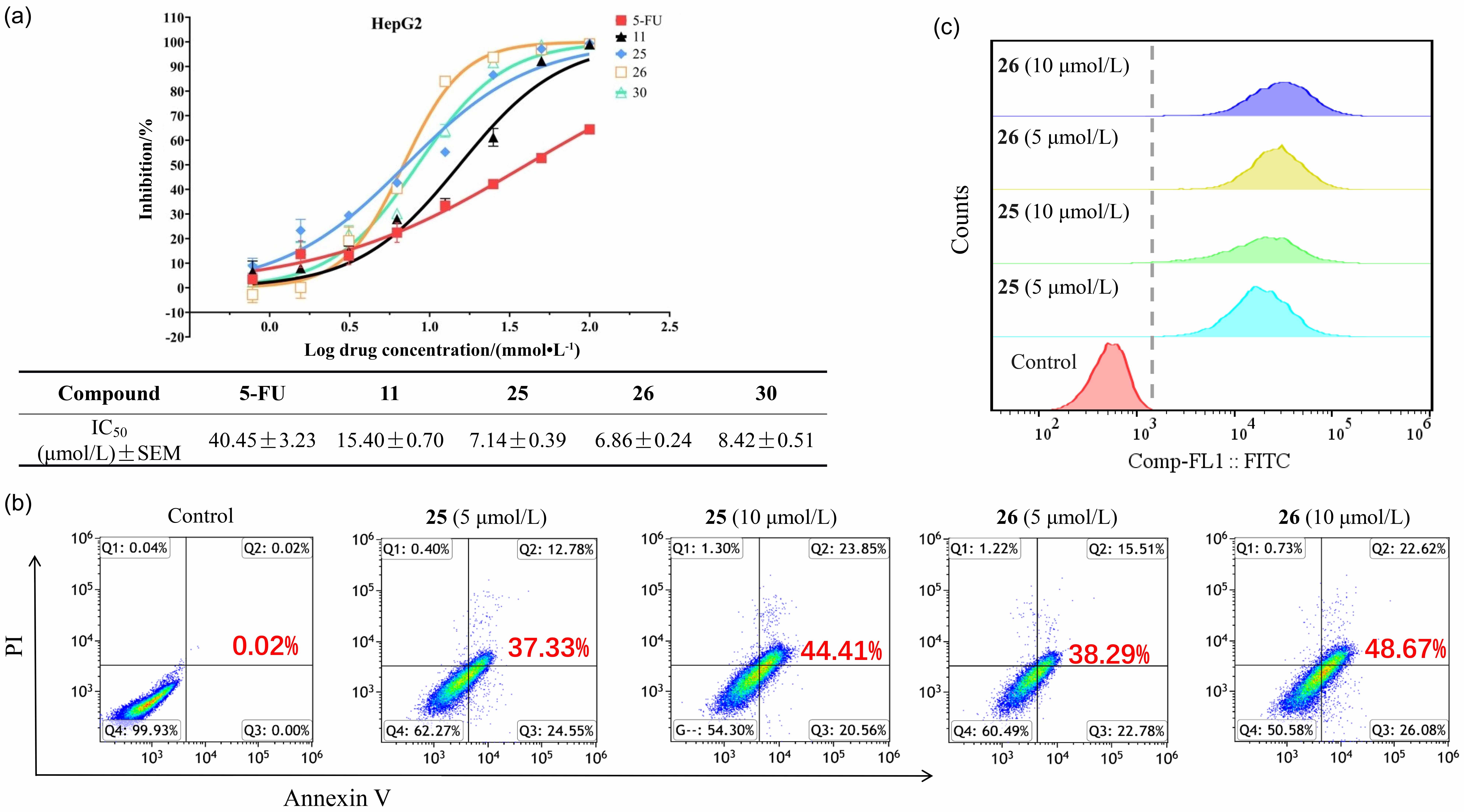
报道了一种镍催化的炔基与三氟乙酰亚胺氯和水的环化反应. 该反应可高效地构建结构多样的2-(三氟甲基)吲哚衍生物. 此策略具有广泛的底物普适性和优异的官能团兼容性. 值得注意的是, 产物(2,5-双(三氟甲基)-1H-吲哚-3-基)(苯基)甲酮(25)和(5-甲基-2-三氟甲基-1H-吲哚-3-基)(苯基)甲酮(26)对HepG2(人肝癌细胞)具有显著的抗增殖活性.
孙雨, 朱延涛, 张东, 倪春节. 镍催化炔基与三氟乙酰亚胺氯和水的环化反应: 高效合成2-(三氟甲基)吲哚衍生物[J]. 有机化学, 2026, 46(3): 1017-1026.
Yu Sun, Yantao Zhu, Dong Zhang, Chunjie Ni. Nickel-Catalyzed Annulation of Alkynyl with Trifluoroacetimidoyl Chlorides and H2O: Facile Access to 2‑(Trifluoromethyl)indole Derivatives[J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 1017-1026.
| Entry | [Ni] | Ligand | Yieldb/% |
|---|---|---|---|
| 1 | NiBr2 | L1 | 62 |
| 2 | NiBr2(dme) | L1 | 61 |
| 3 | NiCl2 | L1 | 56 |
| 4 | Ni(ClO4)2•6H2O | L1 | 58 |
| 5 | Ni(OTf)2 | L1 | 69 |
| 6 | Ni(acac)2 | L1 | 71 |
| 7 | Ni(acac)2 | L2 | 68 |
| 8 | Ni(acac)2 | L3 | 70 |
| 9 | Ni(acac)2 | L4 | 65 |
| 10 | Ni(acac)2 | Xantphos | NDc |
| 11 | Ni(acac)2 | dppBz | 81 |
| 12 | Ni(acac)2 | DPPE | 89 |
| 13 | Ni(acac)2 | DPPP | 72 |
| 14 | Ni(acac)2 | DPPF | ND |
| 15 | Ni(acac)2 | DPEphos | ND |
| 16d | Ni(acac)2 | DPPE | 72 |
| 17e | Ni(acac)2 | DPPE | 48 |
| 18f | Ni(acac)2 | DPPE | Trace |
| Entry | [Ni] | Ligand | Yieldb/% |
|---|---|---|---|
| 1 | NiBr2 | L1 | 62 |
| 2 | NiBr2(dme) | L1 | 61 |
| 3 | NiCl2 | L1 | 56 |
| 4 | Ni(ClO4)2•6H2O | L1 | 58 |
| 5 | Ni(OTf)2 | L1 | 69 |
| 6 | Ni(acac)2 | L1 | 71 |
| 7 | Ni(acac)2 | L2 | 68 |
| 8 | Ni(acac)2 | L3 | 70 |
| 9 | Ni(acac)2 | L4 | 65 |
| 10 | Ni(acac)2 | Xantphos | NDc |
| 11 | Ni(acac)2 | dppBz | 81 |
| 12 | Ni(acac)2 | DPPE | 89 |
| 13 | Ni(acac)2 | DPPP | 72 |
| 14 | Ni(acac)2 | DPPF | ND |
| 15 | Ni(acac)2 | DPEphos | ND |
| 16d | Ni(acac)2 | DPPE | 72 |
| 17e | Ni(acac)2 | DPPE | 48 |
| 18f | Ni(acac)2 | DPPE | Trace |

| [1] |
(a)
doi: 10.1021/jm4017625 |
|
(b)
doi: 10.1016/j.ejmech.2019.111691 |
|
|
(c)
doi: 10.1039/D0NP00088D |
|
| [2] |
(a)
doi: 10.1002/adsc.v367.6 |
|
(b)
doi: 10.1021/acs.accounts.0c00888 |
|
|
(c)
doi: 10.1039/C8CS00454D |
|
| [3] |
(a)
doi: 10.1021/cr040639b |
|
(b)
doi: 10.1021/cr100403z |
|
|
(c)
doi: 10.1039/c2cs15350e |
|
|
(d)
doi: 10.1039/C9QO01315F |
|
|
(e)
doi: 10.1021/acs.accounts.9b00246 |
|
|
(f)
doi: 10.1039/D4CC03040K |
|
|
(g)
doi: 10.1039/D5CC00655D |
|
| [4] |
(a)
doi: 10.1021/acs.chemrev.5b00392 pmid: 17901324 |
|
(b)
doi: 10.1021/acs.jmedchem.9b01877 pmid: 17901324 |
|
|
(c)
doi: 10.1126/science.1131943 pmid: 17901324 |
|
|
(d)
doi: 10.1021/jm800219f pmid: 17901324 |
|
|
(e)
doi: 10.1021/acs.jmedchem.7b01788 pmid: 17901324 |
|
| [5] |
doi: 10.1021/jo00053a011 |
| [6] |
(a)
doi: 10.1039/C9QO01167F |
|
(b)
doi: 10.1039/D3CC00836C |
|
| [7] |
doi: 10.1039/C5RA05967D |
| [8] |
doi: 10.1021/acs.orglett.5c01642 |
| [9] |
(a)
doi: 10.1038/nature13274 |
|
(b)
doi: 10.1002/cjoc.v42.2 |
|
|
(c)
doi: 10.1002/ejoc.v27.46 |
|
|
(d)
doi: 10.1021/acs.accounts.4c00614 |
|
|
(e)
doi: 10.1021/acs.accounts.2c00771 |
|
| [10] |
(a)
doi: 10.1021/acs.jmedchem.9b01218 pmid: 31904232 |
|
(b)
pmid: 31904232 |
|
|
(c)
doi: 10.1016/j.ejmech.2016.06.016 pmid: 31904232 |
|
| [11] |
(a)
doi: 10.1021/acscatal.6b01001 |
|
(b)
doi: 10.1039/D3OB01679J |
|
|
(c)
doi: 10.1021/acs.accounts.5c00013 |
|
| [12] |
doi: 10.6023/cjoc202504001 |
|
(高根伟, 李震, 李炎, 陆熹, 有机化学, 2025, 45, 1905.)
doi: 10.6023/cjoc202504001 |
| [1] | 印丘梅, 刘佳乐, 邓世强, 黄嘉明, 蒋洁, 唐裕才. 锰促进活化烯烃与亚磷酸酯串联磷酰化/环化反应合成γ-咔啉酮衍生物[J]. 有机化学, 2026, 46(3): 1078-1086. |
| [2] | 韦翠, 刘金秋, 邓灿, 邹宁, 周文俊. 螺芴基化合物的合成研究进展[J]. 有机化学, 2026, 46(3): 817-839. |
| [3] | 黄泽宁, 左凯丞, 胡涛, 张澄阳, 张金龙, 蒋高喜. 膦酸催化的双/三次Pictet-Spengler缩合/氧化芳构化构建C2和C3对称的喹啉酮[J]. 有机化学, 2026, 46(3): 1070-1077. |
| [4] | 马超, 徐晓玲, 骆翔, 鲁瑞娟, 虞国棋, 蔡涛. 电化学促进邻炔基苯甲酸酯与硫酚的自由基串联环化反应合成4-硫代异香豆素[J]. 有机化学, 2026, 46(3): 1039-1049. |
| [5] | 周可, 王泽涌, 黎亚琳, 张倩, 李馨月, 邹汉芳, 张明忠, 郭圣荣, 陈铁桥. 一种由四卤化碳生成三氯甲基自由基的氧化策略: 无过渡金属合成三氯甲基化吲哚酮[J]. 有机化学, 2026, 46(3): 974-985. |
| [6] | 何雨蒙, 周风院, 江楠, 张玉琦, 王记江, 马豪杰. 碘催化分子内环化2-羟基-吲哚-3-酮类化合物的合成[J]. 有机化学, 2026, 46(3): 961-967. |
| [7] | 张云一, 严含冰, 朱先进, 石永佳, 李俊鑫, 杨道山, 李旭锋. 镍催化烯烃与环硫鎓盐的还原串联芳烷基化反应[J]. 有机化学, 2026, 46(2): 653-663. |
| [8] | 程文富, 赵明豪, 杨宇晨, 王丽佳. N-Methyl Pratensilin B分子的首次全合成[J]. 有机化学, 2026, 46(2): 670-677. |
| [9] | 郭闻涛, 孟书玉, 王全瑞. 炔基亚砜的分子内交叉偶联/[3,3]-硫鎓离子重排策略构建环烷并[c]呋喃[J]. 有机化学, 2026, 46(2): 594-602. |
| [10] | 曾依玲, 梁方鹏, 李辉, 刘荣荣, 李世清. α-氧代羧酸、炔和伯胺的多组分碳氢环化构筑异喹啉鎓骨架[J]. 有机化学, 2026, 46(2): 554-563. |
| [11] | 罗艳, 刘章伟, 胡朝蕾, 闭红艳, 莫冬亮. 十元氮杂环化合物合成最新研究进展[J]. 有机化学, 2026, 46(2): 420-442. |
| [12] | 高山, 解鑫, 刘元红. 镍/可见光协同催化的溴代芳烃与醇的C—O键偶联反应[J]. 有机化学, 2026, 46(1): 266-278. |
| [13] | 梁媛, 桂超, 王雯雯, 褚雪强, 徐浩, 沈志良. 镍催化的芳基全氟丁基磺酸酯通过碳-氧键官能化的硼化反应高效合成芳基硼酸酯[J]. 有机化学, 2026, 46(1): 207-214. |
| [14] | 蔡压男, 孟祥太. 4-二甲氨基吡啶(DMAP)促进的异喹啉两性离子与α-溴代苯乙酮的[5+1]环化反应: 氢化1,4-噻嗪并异喹啉三环衍生物的高效合成[J]. 有机化学, 2025, 45(9): 3429-3440. |
| [15] | 赵玲, 朱小慧, 陈华, 郑学丽, 薛卫超, 徐嘉麒, 付海燕, 李瑞祥. 钌催化邻硝基苯甲醇与苄胺的转移氢化/环化反应合成喹唑啉[J]. 有机化学, 2025, 45(8): 2836-2847. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||