有机化学 ›› 2026, Vol. 46 ›› Issue (5): 1795-1812.DOI: 10.6023/cjoc202511025 下一篇
综述与进展
收稿日期:2025-11-27
修回日期:2026-01-12
发布日期:2026-02-11
基金资助:
Yun Liao, Lijuan Tang, Yingtong Zong*( ), Daohong Yu*(
), Daohong Yu*( )
)
Received:2025-11-27
Revised:2026-01-12
Published:2026-02-11
Contact:
* E-mail: yudh@gnnu.edu.cn;
zongyingtong@gnnu.edu.cn
Supported by:文章分享
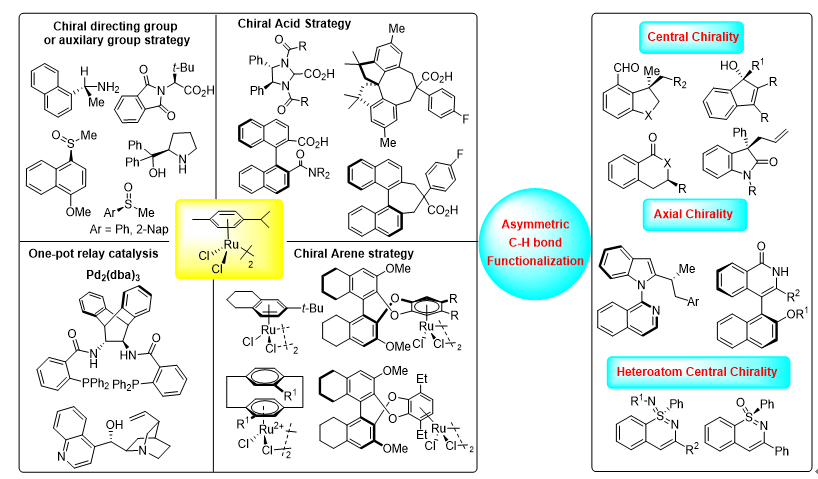
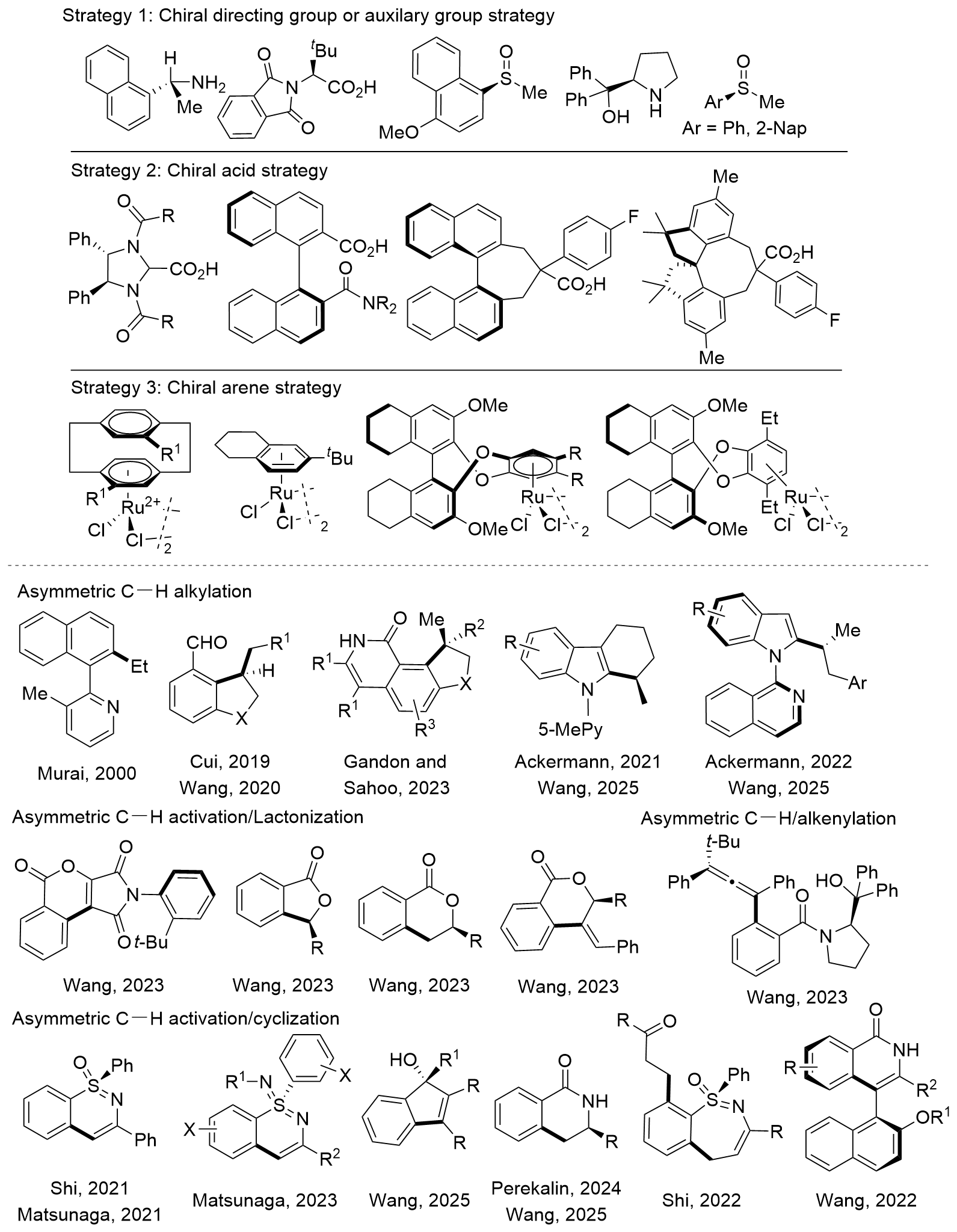
C—H键活化具有无需预官能团化、简洁、高效和绿色等优点, 已成为有机合成中的一种常用策略. 相比于钯、铑和铱等贵金属, 钌催化剂具有催化剂成本低廉、合成容易及稳定好等优点, 使其在C—H活化方面极具前景, 钌催化的不对称C—H键官能团化研究为结构多样的手性化合物的合成提供了新选择. 以不对称C(sp2)—H官能团化中调控产物手性的策略为分类依据, 综述了近年来在钌催化C(sp2)—H键的不对称官能团化领域的进展, 对反应的适用性、官能团兼容性、反应机理和局限性进行了总结, 对该领域所面临的问题和挑战进行了探讨, 并对未来发展方向进行了展望.
廖云, 唐丽娟, 宗映彤, 于道鸿. 钌催化C(sp²)—H键的不对称官能化研究进展[J]. 有机化学, 2026, 46(5): 1795-1812.
Yun Liao, Lijuan Tang, Yingtong Zong, Daohong Yu. Recent Progress in Ruthenium-Catalyzed Asymmetric Functionalization of C(sp²)—H Bonds[J]. Chinese Journal of Organic Chemistry, 2026, 46(5): 1795-1812.

| [1] |
(a)
pmid: 37163671 |
|
(b)
pmid: 37163671 |
|
|
(c)
pmid: 37163671 |
|
|
(d)
pmid: 37163671 |
|
|
(e)
pmid: 37163671 |
|
|
(f)
pmid: 37163671 |
|
|
(g)
pmid: 37163671 |
|
|
(h)
doi: 10.1038/s43586-021-00041-2 pmid: 37163671 |
|
|
(i)
doi: 10.1021/acs.chemrev.2c00888 pmid: 37163671 |
|
|
(j)
doi: 10.6023/A24040136 pmid: 37163671 |
|
|
(黄佳鑫, 刘敏, 徐辉, 戴辉雄, 化学学报, 2024, 82, 565.)
doi: 10.6023/A24040136 pmid: 37163671 |
|
|
(k)
doi: 10.6023/A25010005 pmid: 37163671 |
|
|
(徐安佗, 李骏一, 刘强, , 2025, 83, 390.)
pmid: 37163671 |
|
| [2] |
(a)
doi: 10.1021/acs.chemrev.8b00507 |
|
(b)
|
|
|
(c)
doi: 10.1038/s41586-024-08362-4 |
|
|
(d)
doi: 10.1021/acs.accounts.9b00510 |
|
|
(e)
doi: 10.1246/bcsj.20200349 |
|
|
(f)
doi: 10.1016/j.tetlet.2025.155895 |
|
| [3] |
doi: 10.1021/ja909571z |
| [4] |
(a)
|
|
(b)
doi: 10.1021/acs.chemrev.3c00149 |
|
|
(c)
doi: 10.1039/D3CS00762F |
|
| [5] |
(a)
doi: 10.1016/j.trechm.2021.12.005 |
|
(b)
|
|
| [6] |
(a)
doi: 10.1126/science.1226938 |
|
(b)
doi: 10.1002/anie.v52.40 |
|
|
(c)
doi: 10.1021/ja311956k |
|
|
(d)
doi: 10.1002/ejoc.v2020.42 |
|
| [7] |
doi: 10.1016/j.trechm.2022.01.005 |
| [8] |
(a)
doi: 10.1002/anie.v61.38 |
|
(b)
doi: 10.1002/anie.v61.25 |
|
| [9] |
(a)
doi: 10.1021/acscatal.2c02691 |
|
(b)
doi: 10.1021/jacs.9b02569 |
|
| [10] |
(a)
doi: 10.1038/366529a0 |
|
(b)
doi: 10.1021/ol102942w |
|
| [11] |
(a)
pmid: 11483065 |
|
(b)
doi: 10.1021/ol025851l pmid: 11483065 |
|
| [12] |
(a)
doi: 10.1021/ja710276x |
|
(b)
doi: 10.1039/c1gc15875a |
|
| [13] |
doi: 10.1021/ol201563r pmid: 21749083 |
| [14] |
doi: 10.1021/ol202580e |
| [15] |
(a)
|
|
(b)
|
|
|
(c)
doi: 10.1039/C8CS00036K |
|
|
(d)
|
|
|
(e)
doi: 10.1002/cctc.v16.8 |
|
|
(f)
doi: 10.1016/j.ccr.2020.213602 |
|
|
(g)
|
|
|
(洪朝国, 肖顺丽, 杨凯, 夏家涛, 刘兴旺, 单申, 吴高荣, 化学学报, 2025, 83, 1349.)
doi: 10.6023/A25060237 |
|
| [16] |
(a)
doi: 10.1002/asia.v17.24 |
|
(b)
doi: 10.1055/a-2167-8298 |
|
|
(c)
doi: 10.1360/SSC-2024-0245 |
|
|
(陈文坤, 汪君, 中国科学: 化学, 2025, 55, 370.)
|
|
| [17] |
doi: 10.1002/chem.v29.7 |
| [18] |
(a)
doi: 10.1002/ejoc.v2020.8 |
|
(b)
|
|
|
(c)
doi: 10.1021/acscatal.4c06504 |
|
|
(d)
doi: 10.1021/acs.accounts.3c00081 |
|
|
(e)
doi: 10.1002/chem.v31.12 |
|
| [19] |
|
| [20] |
doi: 10.1021/jacs.9b07251 |
| [21] |
|
| [22] |
doi: 10.1002/anie.v59.9 |
| [23] |
doi: 10.1002/anie.v63.6 |
| [24] |
doi: 10.1021/acs.orglett.3c02969 |
| [25] |
doi: 10.1021/acs.orglett.4c03524 |
| [26] |
doi: 10.1021/cr100412j pmid: 21391562 |
| [27] |
doi: 10.1021/acs.orglett.1c00615 |
| [28] |
doi: 10.1002/anie.v61.47 |
| [29] |
doi: 10.1021/jacs.1c03111 |
| [30] |
doi: 10.1055/a-1588-0072 |
| [31] |
doi: 10.1021/acscatal.2c03531 |
| [32] |
doi: 10.1002/anie.v62.29 |
| [33] |
doi: 10.1002/anie.v61.26 |
| [34] |
(a)
doi: 10.1039/D4CC00181H |
|
(b)
doi: 10.1039/D5DT00344J |
|
| [35] |
doi: 10.1002/anie.v63.31 |
| [36] |
doi: 10.1021/acscatal.4c04798 |
| [37] |
doi: 10.1021/jacs.5c13928 |
| [38] |
doi: 10.1021/jacs.5c20392 |
| [39] |
pmid: 27632781 |
| [40] |
doi: 10.1021/acs.joc.1c02961 |
| [1] | 戴乐薇, 王建玲, 邹东. 芳胺对位C(sp2)—H键官能团化研究进展[J]. 有机化学, 2026, 46(5): 1813-1844. |
| [2] | 白燕茹, 周来运, 刘广华, 王青. 有机大共轭分子C—H亲核取代反应的研究进展[J]. 有机化学, 2026, 46(5): 1883-1896. |
| [3] | 罗辉, 王文权, 贺瑜, 王富强, 杨金会. 无金属电化学C—H键活化构建C—N/C—O键的研究进展[J]. 有机化学, 2026, 46(3): 697-724. |
| [4] | 曾依玲, 梁方鹏, 李辉, 刘荣荣, 李世清. α-氧代羧酸、炔和伯胺的多组分碳氢环化构筑异喹啉鎓骨架[J]. 有机化学, 2026, 46(2): 554-563. |
| [5] | 陈明, 张敬. 芳基羧酸无痕导向官能化反应的研究进展[J]. 有机化学, 2025, 45(8): 2660-2676. |
| [6] | 沈健, 王彦博, 吴正兴, 徐德锋, 张万斌. α,β-不饱和γ-内酯(酰胺)的不对称催化合成[J]. 有机化学, 2025, 45(8): 2637-2659. |
| [7] | 杜一鸣, 贾均松, 李玉龙, 舒伟. 手性α-芳基酮的催化合成研究进展[J]. 有机化学, 2025, 45(6): 1838-1870. |
| [8] | 苏雷, 杨熙, 闫捷, 蒋元力, 陈丽娟, 郑庆舒, 刘家旺. 不对称羰基化偶联反应研究进展[J]. 有机化学, 2025, 45(6): 2007-2047. |
| [9] | 王霜, 毛羊杰, 娄绍杰, 许丹倩. 基于氧化型导向基团的不对称C—H键官能团化反应研究进展[J]. 有机化学, 2025, 45(6): 1961-1994. |
| [10] | 周强, 杨宝臻, 郝贵林, 罗木鹏, 曹石, 赵蓓, 袁华, 王守国. 铑(III)催化的非活化烯烃与α-重氮羰基化合物的对映选择性烯丙位C—H键烷基化反应[J]. 有机化学, 2025, 45(6): 2109-2120. |
| [11] | 唐梦瑶, 杨晓瑜. 手性磷酸催化不对称亲电胺化反应研究进展[J]. 有机化学, 2025, 45(6): 1785-1818. |
| [12] | 蒋晨阳, 尹艳丽, 江智勇. 光酶催化不对称自由基加成反应研究进展[J]. 有机化学, 2025, 45(5): 1614-1633. |
| [13] | 乔秀秀, 李倩, 赵世娜, 魏瑞琪, 马桃, 何永辉, 赵晓静. 2-取代的3H-吲哚-3-酮类化合物参与的C2位手性吲哚啉-3-酮类化合物的不对称合成研究进展[J]. 有机化学, 2025, 45(4): 1166-1177. |
| [14] | 杨之同, 宋恒谦, 雷盼, 闫嘉航, 谢卫青. Communesin生物碱核心五环骨架的高效合成[J]. 有机化学, 2025, 45(3): 1003-1008. |
| [15] | 张朝威, 徐兵斌, 刘文龙, 赵敬, 段伟良. 钯催化不对称碳氢键活化合成平面手性二茂铁磺酰胺化合物[J]. 有机化学, 2025, 45(2): 707-716. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||