有机化学 ›› 2026, Vol. 46 ›› Issue (3): 1060-1069.DOI: 10.6023/cjoc202507034 上一篇 下一篇
研究论文
李乔莉a,†, 王华斌a,†, 覃燕a, 赵加敏a, 韩芳a, 刘雄伟a, 潘博文a, 黄强b,*( ), 周英a,c,*(
), 周英a,c,*( )
)
收稿日期:2025-10-08
修回日期:2025-11-01
发布日期:2025-11-19
作者简介:†共同第一作者
基金资助:
Qiaoli Lia, Huabin Wanga, Yan Qina, Jiamin Zhaoa, Fang Hana, Xiongwei Liua, Bowen Pana, Qiang Huangb,*( ), Ying Zhoua,c,*(
), Ying Zhoua,c,*( )
)
Received:2025-10-08
Revised:2025-11-01
Published:2025-11-19
Contact:
*E-mail: About author:†The authors contributed equally to this work.
Supported by:文章分享
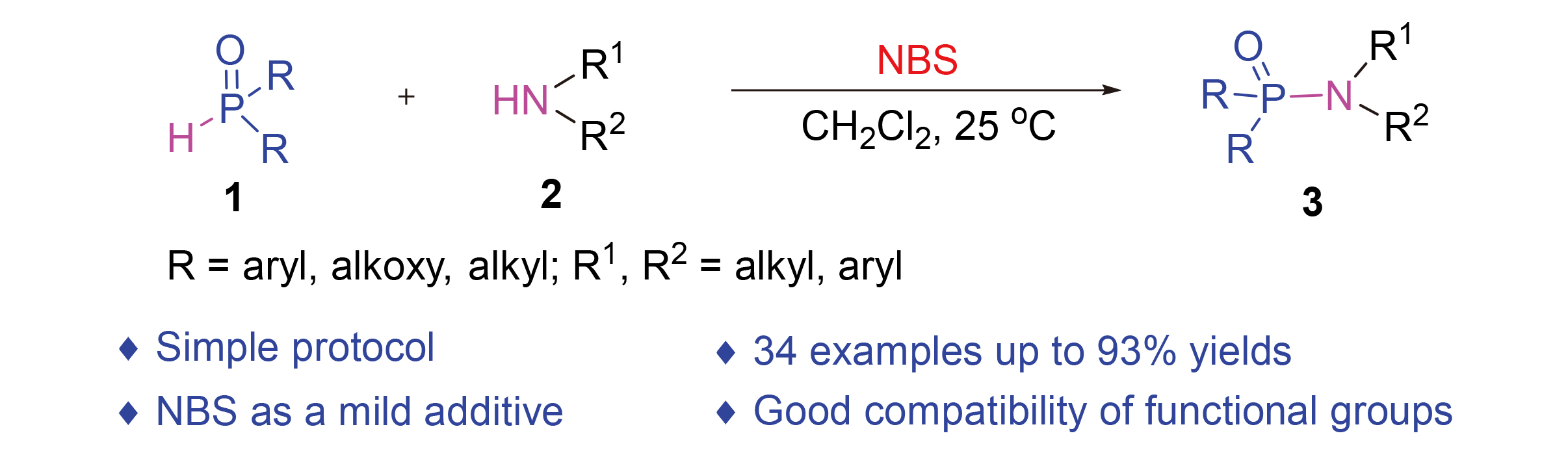
发展了一种温和、绿色、高原子经济性、便捷和可扩大的N-溴代丁二酰亚胺(NBS)促进的次级膦氧化合物与胺的直接磷酰化策略, 用于合成多种磷酰胺类化合物. 底物适用范围广泛且耐受性好, 能以中等至优异的收率(高达93%)合成目标化合物. 该反应在空气中和室温下进行, 无需添加金属催化剂、碱和高的反应温度, 为磷酰胺衍生物的合成提供了一种新的策略.
李乔莉, 王华斌, 覃燕, 赵加敏, 韩芳, 刘雄伟, 潘博文, 黄强, 周英. N-溴代丁二酰亚胺促进的P(O)—H化合物参与的胺的直接磷酰化反应[J]. 有机化学, 2026, 46(3): 1060-1069.
Qiaoli Li, Huabin Wang, Yan Qin, Jiamin Zhao, Fang Han, Xiongwei Liu, Bowen Pan, Qiang Huang, Ying Zhou. N-Bromosuccinimide-Promoted Direct Phosphorylation of Amines with P(O)—H Compounds[J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 1060-1069.
| Entry | Additive | Solvent | T/℃ | Yieldb/% |
|---|---|---|---|---|
| 1 | NBS | Toluene | 25 | 65 |
| 2 | NCS | Toluene | 25 | 62 |
| 3 | NIS | Toluene | 25 | 59 |
| 4 | NBS | CH3CN | 25 | 76 |
| 5 | NBS | DMF | 25 | None |
| 6 | NBS | DMSO | 25 | None |
| 7 | NBS | DCM | 25 | 93 |
| 8 | NBS | EA | 25 | 65 |
| 9 | NBS | Dioxane | 25 | 57 |
| 10 | NBS | CS2 | 25 | 75 |
| 11 | NBS | Chloroform | 25 | 87 |
| 12 | NBS | CCl4 | 25 | 85 |
| 13 | NBS | TH4 | 25 | 67 |
| 14 | NBS | Acetone | 25 | 65 |
| 15 | NBS | DCM | 0 | 78 |
| 16 | NBS | DCM | 45 | 87 |
| 17 | NBS | DCM | 60 | 76 |
| 18 | NBS | DCM | 80 | 65 |
| 19 | NBS | DCM | 25 | 45c, 64d, 72e |
| Entry | Additive | Solvent | T/℃ | Yieldb/% |
|---|---|---|---|---|
| 1 | NBS | Toluene | 25 | 65 |
| 2 | NCS | Toluene | 25 | 62 |
| 3 | NIS | Toluene | 25 | 59 |
| 4 | NBS | CH3CN | 25 | 76 |
| 5 | NBS | DMF | 25 | None |
| 6 | NBS | DMSO | 25 | None |
| 7 | NBS | DCM | 25 | 93 |
| 8 | NBS | EA | 25 | 65 |
| 9 | NBS | Dioxane | 25 | 57 |
| 10 | NBS | CS2 | 25 | 75 |
| 11 | NBS | Chloroform | 25 | 87 |
| 12 | NBS | CCl4 | 25 | 85 |
| 13 | NBS | TH4 | 25 | 67 |
| 14 | NBS | Acetone | 25 | 65 |
| 15 | NBS | DCM | 0 | 78 |
| 16 | NBS | DCM | 45 | 87 |
| 17 | NBS | DCM | 60 | 76 |
| 18 | NBS | DCM | 80 | 65 |
| 19 | NBS | DCM | 25 | 45c, 64d, 72e |
| [1] |
(a)
doi: 10.1016/j.polymer.2021.123491 |
|
(b)
|
|
|
(c)
doi: 10.1002/adsc.v362.12 |
|
| [2] |
(a)
doi: 10.1016/j.molstruc.2021.130528 pmid: 28124918 |
|
(b)
doi: 10.1039/D1OB01566D pmid: 28124918 |
|
|
(c)
doi: 10.3389/fmicb.2020.02045 pmid: 28124918 |
|
|
(d)
doi: 10.1016/j.chemosphere.2020.126549 pmid: 28124918 |
|
|
(e)
doi: 10.1016/j.rechem.2022.100346 pmid: 28124918 |
|
|
(f)
pmid: 28124918 |
|
|
(g)
doi: 10.1021/acs.inorgchem.3c04521 pmid: 28124918 |
|
|
(h)
doi: 10.1016/j.rechem.2024.101498 pmid: 28124918 |
|
|
(i)
doi: 10.1002/chem.201700218 pmid: 28124918 |
|
|
(j)
doi: 10.1021/acs.orglett.6b03625 pmid: 28124918 |
|
| [3] |
(a)
doi: 10.1016/j.ejmech.2012.05.017 |
|
(b)
doi: 10.14233/ajchem |
|
| [4] |
(a)
pmid: 24783964 |
|
(b)
doi: 10.1016/j.tet.2013.09.001 pmid: 24783964 |
|
|
(c)
doi: 10.1021/ol500894k pmid: 24783964 |
|
|
(d)
doi: 10.6023/cjoc202210032 pmid: 24783964 |
|
|
(方思强, 刘赞娇, 王天利, 有机化学, 2023, 43, 1069.)
pmid: 24783964 |
|
|
(e)
doi: 10.1080/10426507.2018.1513511 pmid: 24783964 |
|
| [5] |
doi: 10.1055/s-0036-1589111 |
| [6] |
doi: 10.1039/C9CC09407E |
| [7] |
doi: 10.1002/chem.v26.4 |
| [8] |
doi: 10.1039/D4OB01860E |
| [9] |
doi: 10.1002/adsc.v358.5 |
| [10] |
doi: 10.1002/adsc.v363.2 |
| [11] |
pmid: 8917645 |
| [12] |
(a)
doi: 10.1038/s41467-022-31178-7 pmid: 35715392 |
|
(b)
doi: 10.1007/s11426-021-9971-9 pmid: 35715392 |
|
|
(c)
doi: 10.1039/D2GC01513G pmid: 35715392 |
|
|
(d)
doi: 10.31635/ccschem.021.202101615 pmid: 35715392 |
|
|
(e)
doi: 10.1039/D3QO01375H pmid: 35715392 |
|
|
(f)
doi: 10.1002/chem.v26.47 pmid: 35715392 |
|
|
(g)
doi: 10.1021/acs.joc.3c02416 pmid: 35715392 |
|
| [13] |
(a)
doi: 10.1080/00397911.2016.1205196 |
|
(b)
doi: 10.1016/j.tetlet.2017.05.005 |
|
|
(c)
doi: 10.1002/cjoc.v35.9 |
|
|
(d)
doi: 10.1016/j.tetlet.2017.04.042 |
|
|
(e)
doi: 10.1039/D4OB01244E |
|
|
(f)
doi: 10.6023/cjoc20203002 |
|
|
(王华斌, 徐连华, 刘雄伟, 潘博文, 姚震, 黄强, 周英, 有机化学, 2024, 44, 2847.)
|
|
| [14] |
(a)
doi: 10.1021/acscatal.0c00281 pmid: 36540271 |
|
(b)
doi: 10.1021/acs.orglett.0c03030 pmid: 36540271 |
|
|
(c)
pmid: 36540271 |
|
|
(d)
doi: 10.1055/a-1277-8669 pmid: 36540271 |
|
|
(e)
pmid: 36540271 |
|
|
(王化坤, 任晓龙, 宣宜宁, 有机化学, 2024, 44, 251.)
pmid: 36540271 |
|
|
(f)
doi: 10.6023/cjoc202202020 pmid: 36540271 |
|
|
(钱存卫, 韩容, 沈芷欣, 李倩, 陈选荣, 有机化学, 2022, 42, 2496.)
pmid: 36540271 |
|
| [15] |
doi: 10.1016/j.tetlet.2016.05.076 |
| [16] |
(a)
doi: 10.1021/acs.orglett.3c02563 |
|
(b)
doi: 10.1039/C7OB01104K |
|
|
(c)
doi: 10.6023/cjoc201708023 |
|
|
(高玉珍, 唐果, 赵玉芬, 有机化学, 2018, 38, 62.)
|
|
|
(d)
doi: 10.6023/cjoc201702050 |
|
|
(杨佳, 肖晶, 周永波, 陈铁桥, 尹双凤, 韩立彪, 有机化学, 2017, 37, 1055.)
|
|
|
(e)
doi: 10.6023/cjoc201312023 |
|
|
(杨斌, 田秋萍, 杨尚东, 有机化学, 2014, 34, 717.)
|
|
| [17] |
(a)
doi: 10.1021/acs.orglett.9b01081 |
|
(b)
doi: 10.6023/cjoc201805027 |
|
|
(王华斌, 付强, 张智杰, 高明, 姬建新, 易东, 有机化学, 2018, 38, 1977.)
|
|
| [18] |
(a)
doi: 10.1021/acs.orglett.3c03193 |
|
(b)
doi: 10.1021/acs.joc.3c01583 |
|
|
(c)
doi: 10.1021/acs.orglett.3c01939 |
|
|
(d)
doi: 10.1021/acs.joc.3c02716 |
| [1] | 印丘梅, 刘佳乐, 邓世强, 黄嘉明, 蒋洁, 唐裕才. 锰促进活化烯烃与亚磷酸酯串联磷酰化/环化反应合成γ-咔啉酮衍生物[J]. 有机化学, 2026, 46(3): 1078-1086. |
| [2] | 孙雨, 朱延涛, 张东, 倪春节. 镍催化炔基与三氟乙酰亚胺氯和水的环化反应: 高效合成2-(三氟甲基)吲哚衍生物[J]. 有机化学, 2026, 46(3): 1017-1026. |
| [3] | 何雨蒙, 周风院, 江楠, 张玉琦, 王记江, 马豪杰. 碘催化分子内环化2-羟基-吲哚-3-酮类化合物的合成[J]. 有机化学, 2026, 46(3): 961-967. |
| [4] | 杨洁, 佘湫楠, 周逸聪, 康丽琴, 赵佳丽, 刘传祥. 氰基导向芳香氢亲核取代反应构建新型萘酰亚胺比色探针: 食品与水样中氰化物的高灵敏检测[J]. 有机化学, 2026, 46(2): 531-538. |
| [5] | 梁婉婷, 陈思怡, 徐恩琪, 陈雪冰. 铜催化3-硝基色烯与烯胺酮环化反应合成2H-色烯并吡咯衍生物[J]. 有机化学, 2026, 46(2): 496-506. |
| [6] | 苏钰盈, 王映芳, 易梦镯, 赵立兴, 李佳璇, 马玲, 尹敏. 仙鹤草内生链霉菌YINM00048中戊二酰亚胺类衍生物的研究[J]. 有机化学, 2026, 46(1): 289-294. |
| [7] | 谭紫云, 杨新, 龚绍峰, 杨慧翎, 谢永燕, 张静雅, 冯浙泰, 李文艺, 肖新生. 非均相铜催化的叔胺与炔烃需氧氧化交叉偶联反应[J]. 有机化学, 2026, 46(1): 156-166. |
| [8] | 王文龙, 温家旭, 陈飞, 薄春博, 李敏, 刘宁, 杜智宏. 铜催化的3-羟基-2-萘甲酸酯的不对称氧化偶联反应:氨基酸类配体的设计与优化[J]. 有机化学, 2026, 46(1): 167-180. |
| [9] | 周蒲, 苏子洋, 崔得运, 程绎南, 李毅, 周海峰, 孙炳剑, 李洪连. 硫色酮甲酰胺衍生物的合成及其抑菌活性[J]. 有机化学, 2026, 46(1): 87-95. |
| [10] | 杨威, 王兵兵, 徐小惠, 孙钟, 唐翠曼, 刘佳琦. 仿生材料聚多巴胺在有机转化中的应用进展[J]. 有机化学, 2025, 45(9): 3314-3325. |
| [11] | 贾晨旭, 安聪好, 黄军. 铜催化C—O偶联制备间苯氧基苯甲醛[J]. 有机化学, 2025, 45(9): 3412-3419. |
| [12] | 李欢乐, 潘其, 娄绍杰, 毛羊杰, 许丹倩. 基于氢原子迁移(HAT)过程的胺类化合物选择性C—H键转化反应研究进展[J]. 有机化学, 2025, 45(9): 3213-3243. |
| [13] | 汤敏, 张斌, 王秋实, 方超华, 胡立威, 关丽萍. 基于包含吲哚环、苯并噻吩环的单胺氧化酶和胆碱酯酶抑制活性的查尔酮衍生物设计、合成及生物活性研究[J]. 有机化学, 2025, 45(8): 2989-3003. |
| [14] | 刘丽强, 南军, 段兴宇, 王昊龙, 蔡巷, 王梓诚, 张恩泽, 孙彦民. 三齿P配体钌络合物催化脂肪醇胺N-烷基化反应研究[J]. 有机化学, 2025, 45(8): 2983-2988. |
| [15] | 解人杰, 谢复开, 孙然, 王欣, 王钰佳, 李蕾, 王贺. 电子供体-受体复合物(EDA)介导N-芳基丙烯酰胺与芳基硫鎓盐的自由基环化反应[J]. 有机化学, 2025, 45(8): 2913-2922. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||