有机化学 ›› 2026, Vol. 46 ›› Issue (4): 1205-1221.DOI: 10.6023/cjoc202601016 上一篇 下一篇
综述与进展
收稿日期:2026-01-13
修回日期:2026-03-10
发布日期:2026-04-13
通讯作者:
舒超
作者简介:† 共同第一作者
基金资助:
Xinran Hana, Hengyu Huoa, Xuepan Songb, Chao Shua,*( )
)
Received:2026-01-13
Revised:2026-03-10
Published:2026-04-13
Contact:
Chao Shu
About author:†These authors contributed equally to this work
Supported by:文章分享
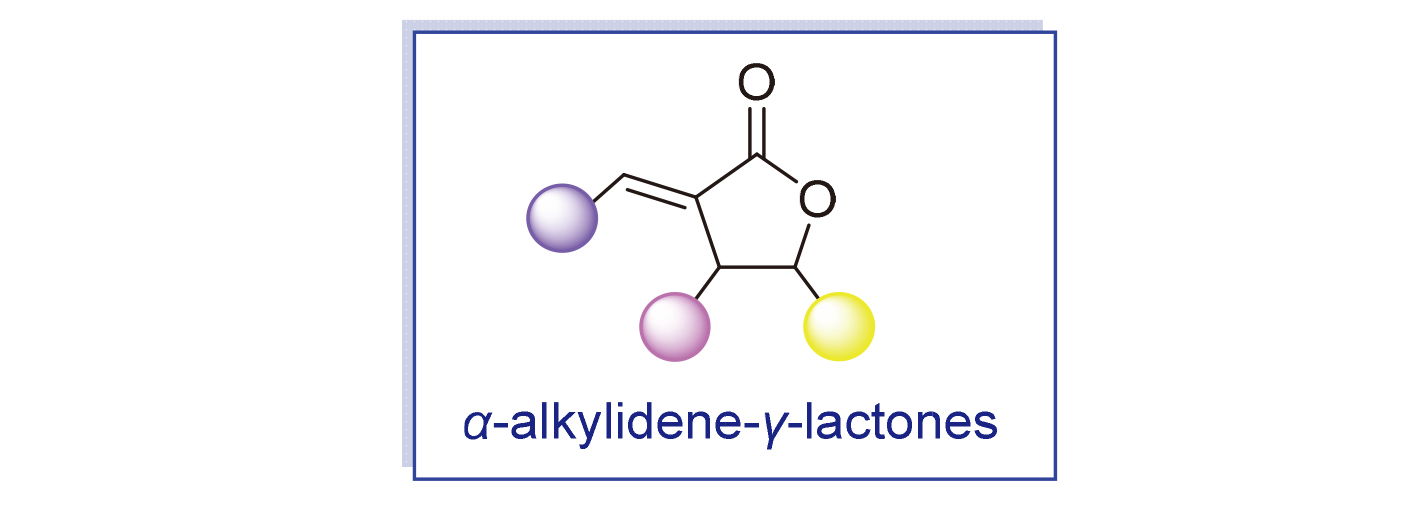
α-烯基-γ-内酯骨架由于具有多种生物活性, 是许多天然产物和活性药物中核心的结构单元. 在过去几年中, α-烯基-γ-内酯的合成研究取得显著进展, 研究者们利用新型合成技术, 发展出了高效且选择性强的催化方法, 这些方法能够在相对温和的条件下实现其合成. 然而, 近十年, 该研究领域仍缺乏系统性的全面综述. 此文介绍了典型的含有α-烯基-γ-内酯的天然和非天然生物活性分子, 并重点综述了α-烯基-γ-内酯衍生物的最新合成进展, 希望能够激发更多的α-烯基-γ-内酯合成新策略, 并促进其在药物研发中的应用.
韩昕冉, 霍恒宇, 宋学攀, 舒超. α-烯基-γ-内酯的合成进展[J]. 有机化学, 2026, 46(4): 1205-1221.
Xinran Han, Hengyu Huo, Xuepan Song, Chao Shu. Recent Advances for the Synthesis of α-Alkylidene-γ-lactones[J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1205-1221.
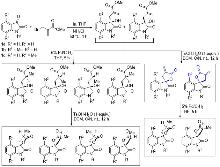
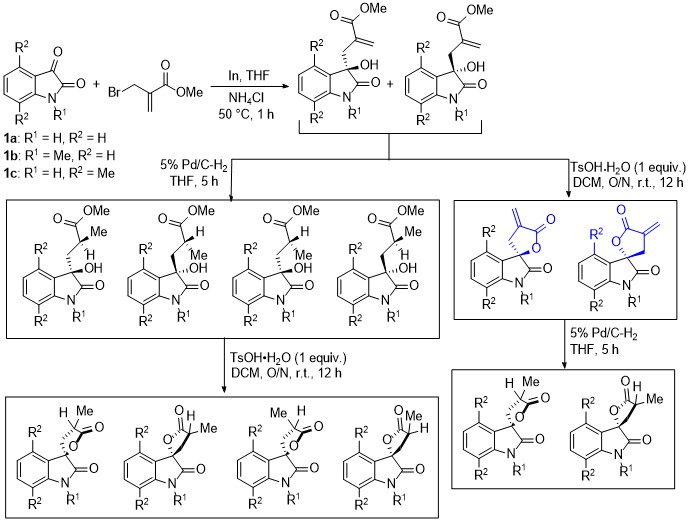

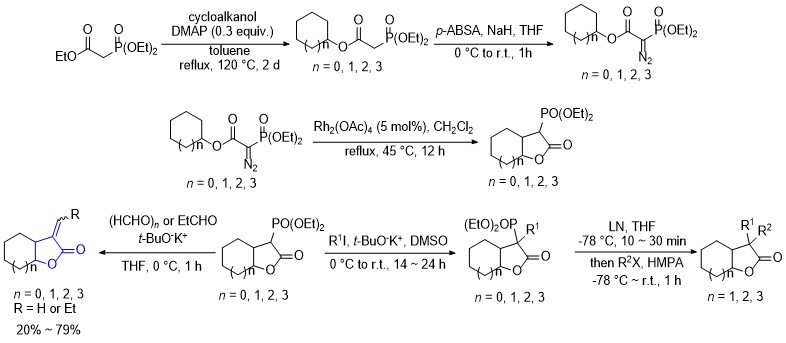
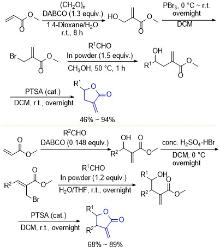
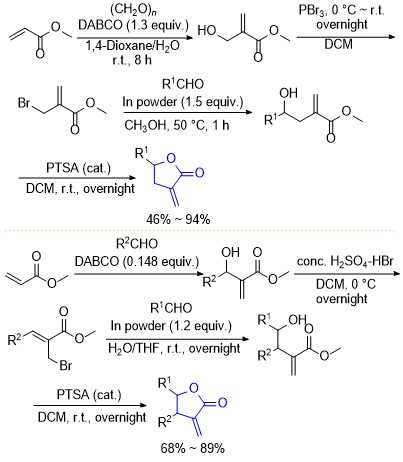
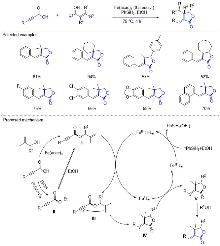
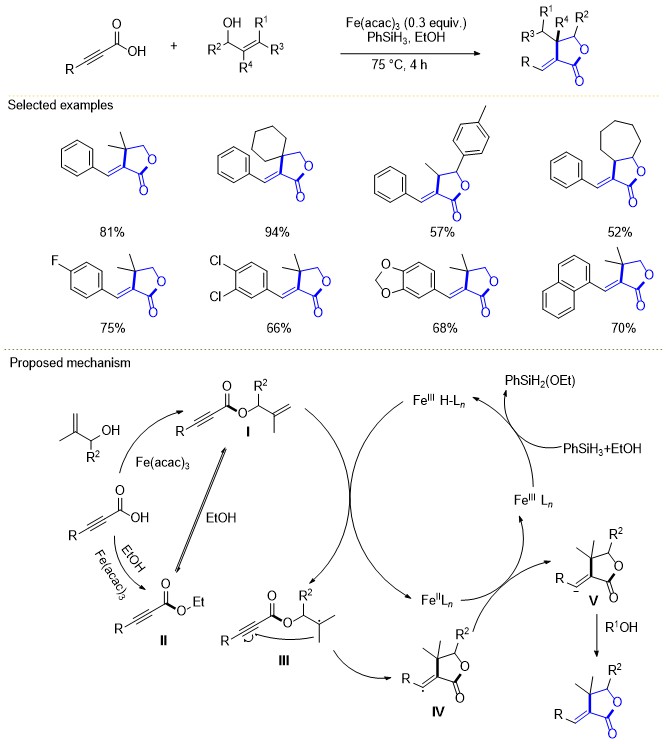
| [1] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
(a)
|
|
(b)
|
|
| [6] |
|
| [7] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [8] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
| [9] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
(a)
|
|
(b)
|
|
| [16] |
|
| [17] |
|
| [18] |
(a)
|
|
(b)
|
|
| [19] |
(a)
|
|
(b)
|
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
(a)
|
|
(b)
|
|
| [41] |
|
| [42] |
(a)
|
|
(b)
|
|
| [43] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
| [44] |
(a)
|
|
(b)
|
|
| [45] |
(a)
|
|
(b)
|
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
|
(杨政, 黄丹凤, 文岚, 王娟娟, 王克虎, 胡雨来, 有机化学, 2018, 38, 1725.)
|
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
|
(李建晓, 张振明, 李春生, 罗维, 杨少容, 有机化学, 2015, 35, 2199.)
|
|
| [58] |
|
| [61] |
|
| [59] |
|
| [60] |
|
| [1] | 邹震雷, 李益凡, 陈晨, 陈王哲, 张为钢, 王毅. 关于亚磺酰基砌块法合成亚砜的研究进展[J]. 有机化学, 2026, 46(4): 1635-1658. |
| [2] | 唐玉青, 杨紫娴, 杨兵, 王毅, 杜炳南. 球磨条件下机械力驱动的自由基反应研究进展[J]. 有机化学, 2026, 46(4): 1111-1145. |
| [3] | 孟令旭, 申霖, 罗祥玲, 林玉妹, 龚磊. 具有光活性的铬、钴配合物的设计及其在有机合成中的应用进展[J]. 有机化学, 2026, 46(4): 1513-1528. |
| [4] | 马超, 徐晓玲, 骆翔, 鲁瑞娟, 虞国棋, 蔡涛. 电化学促进邻炔基苯甲酸酯与硫酚的自由基串联环化反应合成4-硫代异香豆素[J]. 有机化学, 2026, 46(3): 1039-1049. |
| [5] | 周顺丽, 刘继, 雷婷, 杨斌全, 江世智. 2-芳基-2H-色烯类化合物的手性合成研究进展[J]. 有机化学, 2026, 46(3): 773-785. |
| [6] | 王雪倩, 段宝忠, 田红畅, 张磊. 吲哚生物碱Catharanthine合成研究进展[J]. 有机化学, 2026, 46(3): 725-742. |
| [7] | 张涛, 季珍瑜, 唐功利, 侯现锋. 金色链霉菌SP-371中一个新型倍半萜内酯化合物的发现[J]. 有机化学, 2026, 46(2): 689-692. |
| [8] | 程文富, 赵明豪, 杨宇晨, 王丽佳. N-Methyl Pratensilin B分子的首次全合成[J]. 有机化学, 2026, 46(2): 670-677. |
| [9] | 何卫刚, 段明月, 闫帅, 叶伟杰, 徐雪园, 孙先宇. 蕊木属生物碱arboridinine和arborisidine的全合成研究进展[J]. 有机化学, 2026, 46(2): 443-454. |
| [10] | 赵咪娜, 唐嘉一. 咪唑类化合物的合成研究进展[J]. 有机化学, 2026, 46(2): 455-474. |
| [11] | 郭峻烽, 李春迎, 胡荣贵, 戎豪杰, 李江伟, 杜咏梅, 秦越, 吕剑, 孙道安. 环丙烷骨架的构建及其应用研究进展[J]. 有机化学, 2026, 46(2): 379-398. |
| [12] | 田灵燕, 王守锋, 曾伟. 大蒜中S-1-丙烯基-L-半胱氨酸的合成及生物学特性研究进展[J]. 有机化学, 2026, 46(1): 74-86. |
| [13] | 赵礼麟, 李兆麒, 钟李平, 闵龙. abeo-甾体的全合成研究进展[J]. 有机化学, 2026, 46(1): 21-38. |
| [14] | 崔心怡, 郭丽帆, 马聪璇, 李耘, 梁建华. 抗耐药菌红霉素的结构修饰策略、构效关系及全合成研究进展[J]. 有机化学, 2026, 46(1): 39-73. |
| [15] | 周蒲, 苏子洋, 崔得运, 程绎南, 李毅, 周海峰, 孙炳剑, 李洪连. 硫色酮甲酰胺衍生物的合成及其抑菌活性[J]. 有机化学, 2026, 46(1): 87-95. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||