Chinese Journal of Organic Chemistry ›› 2026, Vol. 46 ›› Issue (4): 1464-1480.DOI: 10.6023/cjoc202512007 Previous Articles Next Articles
REVIEWS
收稿日期:2025-12-05
修回日期:2026-01-07
发布日期:2026-03-06
通讯作者:
董建洋, 薛东
基金资助:Received:2025-12-05
Revised:2026-01-07
Published:2026-03-06
Contact:
Jianyang Dong, Dong Xue
Supported by:Share
Jianyang Dong, Dong Xue. Research Progress on the Functionalization of [1.1.1]Propellane via Radical Pathways[J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1464-1480.
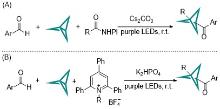
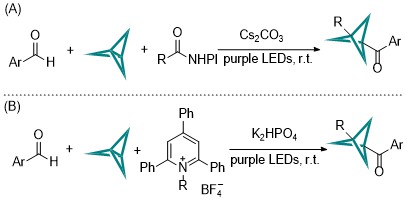
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
| [72] |
|
| [73] |
|
| [74] |
|
|
(张红, 王明扬, 吴新鑫, 朱晨, 有机化学, 2020, 40, 3431 )
|
|
| [75] |
|
| [76] |
|
| [77] |
|
| [78] |
|
| [1] | Xinyi Hou, Tongtong Shi, Zejiang Li, Qinghao Dong, Kai Sun, Xin Wang. Visible-Light-Induced Phosphorylation Cyclization to Phosphorylated Indole-Fused Diazepines [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1776-1787. |
| [2] | Zhenlei Zou, Yifan Li, Chen Chen, Wangzhe Chen, Weigang Zhang, Yi Wang. Recent Advances in Sulfoxide Synthesis via Sulfinyl Building Blocks [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1635-1658. |
| [3] | Qixiang Wang, Shengxi Chen, Jing Wang, Dong Chen, Xiaoguang Bao, Xinxin Wu. Metal-Free Selective Chlorination of Polyenes [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1750-1762. |
| [4] | Jingfei Zheng, Qiyan Lv, Kai Sun, Xiaolan Chen, Lingbo Qu, Jinquan Wang, Bing Yu. Recent Advances in Functionalization of 6-Azauracils via Radical Reactions [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1603-1620. |
| [5] | Shizheng Liu, Mingyu Dou, Yu Cui, Jianmin Dou, Wenting Wei. Research Progress on Radical Selenosulfonylation [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1621-1634. |
| [6] | Yuqing Zhao, Xianjin Zhu, Lingyu Shi, Mingyu Wei, Zihan Xu, Yongjia Shi, Xufeng Li, Daoshan Yang. Research Progress on the Ring-Opening Functionalization of Cyclic Sulfonium Salts [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1572-1602. |
| [7] | Haoran Zhang, Junsong Jia, Qiong Yu, Yu Wu, Yulong Li, Wei Shu. Visible-Light-Catalyzed Hydroamination, Hydroamidation and Hydrosulfonamidation of Alkenes [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1166-1180. |
| [8] | Shan Yang, Yasu Chen, Chen Zhu. Construction of N-Fused Heteroarenes via Radical-Mediated Functional Group Migration-Cyclization [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1739-1749. |
| [9] | Jiahao Chen, Beibei Zhan, Xiaheng Zhang. Research Progress in Radical-Mediated 1,n-Nitrogen Migration Reactions [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1560-1571. |
| [10] | Fu Ye, Weiming Yuan. Recent Advances in Dicarbofunctionalization of Alkynes via Photoredox Catalysis [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1146-1165. |
| [11] | Linyu Lu, Yutong Qi, Tinghui Zhang, Shengnan Jin, Zhongyan Cao, Hongwei Zhang. Recent Progress in Radical Mediated 1,2,n-Trifunctiona-lization of Olefins [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1540-1559. |
| [12] | Yuqing Tang, Zixian Yang, Bing Yang, Yi Wang, Bingnan Du. Research Progress in Mechanically Driven Radical Reactions under Ball Milling Conditions [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1111-1145. |
| [13] | Yu-Chen Miao, Hang Qi, Huan-Ming Huang. Recent Advances in Enantioselective Transformations Enabled by Synergistic Photoredox/Chromium Catalysis [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1481-1495. |
| [14] | Xiaoman Wang, Ze Wang, Jie Wu, Yi Hu, Shengqing Ye. Research Advances on Radical Reactions in Aqueous Phase [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1496-1512. |
| [15] | Lingxu Meng, Lin Shen, Xiangling Luo, Yumei Lin, Lei Gong. Design of Photoactive Cr- and Co-Based Complexes and Their Applications in Organic Synthesis [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1513-1528. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
