有机化学 ›› 2026, Vol. 46 ›› Issue (2): 554-563.DOI: 10.6023/cjoc202507007 上一篇 下一篇
研究论文
收稿日期:2025-07-04
修回日期:2025-09-22
发布日期:2025-10-23
通讯作者:
李世清
基金资助:
Yiling Zeng, Fangpeng Liang, Hui Li, Rongrong Liu, Shiqing Li*( )
)
Received:2025-07-04
Revised:2025-09-22
Published:2025-10-23
Contact:
Shiqing Li
Supported by:文章分享
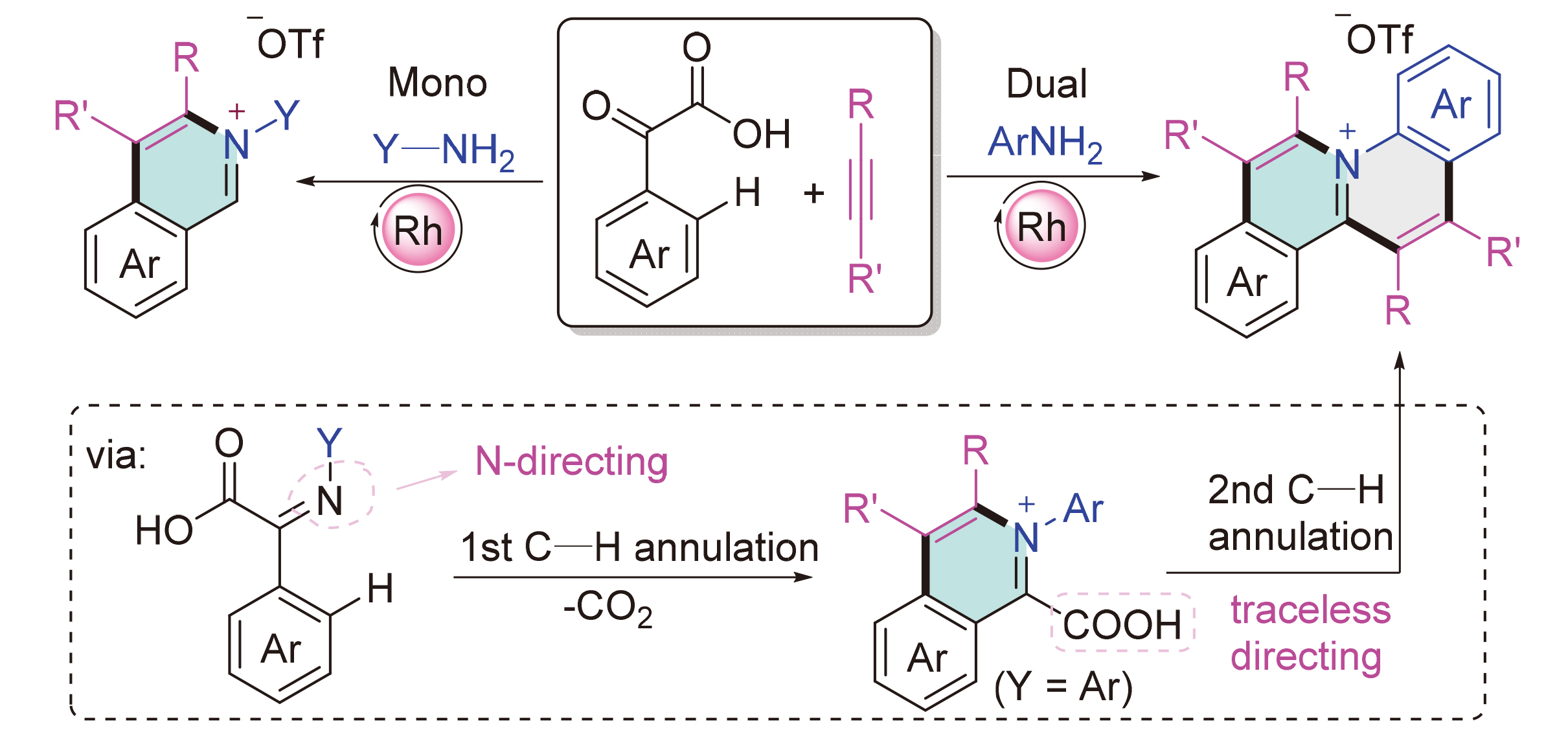
开发了Rh(III)催化α-氧代羧酸、炔和伯胺的三组分C—H键活化/环化反应, 一步构筑二、四并环的异喹啉鎓盐衍生物. 伯胺与α-氧代羧酸通过酮胺缩合原位生成α-亚胺酸, 从而将α-氧代羧酸的弱氧导向转变成强氮导向. 当烷基胺为氮源时, α-亚胺酸与炔烃发生氮导向的C—H键活化/环化反应, 以较高的产率得到二并环的N-烷基异喹啉鎓盐(离子液). 当芳胺作为氮源时, α-亚胺酸与炔烃发生氮导向的C—H环化后, 可以进一步发生羧基导向的C—H键活化/环化反应, 从而以中等至良好的产率得到四并环的二苯并[a,f]喹嗪鎓盐. 通过时间依赖的ESI-MS成功检测到7个关键中间体, 其中包括3个羧基保留的物种, 证明了羧基在该反应中的重要性, 并以此提出包含了酮胺缩合、脱羧、C—H键活化、炔烃插入及还原消除等过程的反应机理.
曾依玲, 梁方鹏, 李辉, 刘荣荣, 李世清. α-氧代羧酸、炔和伯胺的多组分碳氢环化构筑异喹啉鎓骨架[J]. 有机化学, 2026, 46(2): 554-563.
Yiling Zeng, Fangpeng Liang, Hui Li, Rongrong Liu, Shiqing Li. Multi-component C—H Annulation of α-Oxocarboxylic Acids, Allynes and Primary Amines to Yield Isoquinolinium Skeletons[J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 554-563.
| Entry | Variation from above conditionsa | 3a/4ab/% |
|---|---|---|
| 1 | None | 74/trace |
| 2 | [(p-cymene)RuCl2] as catalyst | 0/0 |
| 3 | [Cp*Co(CO)I2] as catalyst | 0/0 |
| 4 | [Cp*RhCl2]2 (2.5 mol%) | 10/47 |
| 5 | Cu(OTf)2 instead of Cu(OAc)2 | 21/0 |
| 6 | AgOAc instead of Cu(OAc)2 | 12/31 |
| 7 | Cu(OAc)2 (2 equiv.) | 26/55 |
| 8 | Without Cu(OAc)2 | 9/56 |
| 9 | Without NaOAc | <5/48 |
| 10 | HOAc instead of NaOAc | 25/51 |
| 11 | PivOH instead of NaOAc | 26/50 |
| 12 | CsOAc instead of NaOAc | 29/36 |
| 13 | NaBF4 instead of Zn(OTf)2 | 66/<5 |
| 14 | NaSbF6 instead of Zn(OTf)2 | 67/<5 |
| Entry | Variation from above conditionsa | 3a/4ab/% |
|---|---|---|
| 1 | None | 74/trace |
| 2 | [(p-cymene)RuCl2] as catalyst | 0/0 |
| 3 | [Cp*Co(CO)I2] as catalyst | 0/0 |
| 4 | [Cp*RhCl2]2 (2.5 mol%) | 10/47 |
| 5 | Cu(OTf)2 instead of Cu(OAc)2 | 21/0 |
| 6 | AgOAc instead of Cu(OAc)2 | 12/31 |
| 7 | Cu(OAc)2 (2 equiv.) | 26/55 |
| 8 | Without Cu(OAc)2 | 9/56 |
| 9 | Without NaOAc | <5/48 |
| 10 | HOAc instead of NaOAc | 25/51 |
| 11 | PivOH instead of NaOAc | 26/50 |
| 12 | CsOAc instead of NaOAc | 29/36 |
| 13 | NaBF4 instead of Zn(OTf)2 | 66/<5 |
| 14 | NaSbF6 instead of Zn(OTf)2 | 67/<5 |
| [1] |
(a)
doi: 10.1039/c3cs60147a pmid: 24077263 |
|
(b)
doi: 10.1039/c2cs35211g pmid: 24077263 |
|
| [2] |
(a)
doi: 10.1021/acs.chemrev.6b00076 pmid: 27258218 |
|
(b)
doi: 10.1021/acs.chemrev.8b00637 pmid: 27258218 |
|
|
(c)
doi: 10.1021/acs.chemrev.1c00449 pmid: 27258218 |
|
| [3] |
(a)
doi: 10.1039/C8SC01963K pmid: 30074773 |
|
(b)
doi: 10.1002/adfm.v30.42 pmid: 30074773 |
|
|
(c)
doi: 10.1021/jacs.7b10150 pmid: 30074773 |
|
|
(d)
doi: 10.1021/acsnano.8b03138 pmid: 30074773 |
|
|
(e)
doi: 10.1002/adfm.v30.10 pmid: 30074773 |
|
|
(f)
doi: 10.1002/anie.v59.24 pmid: 30074773 |
|
| [4] |
(a)
doi: 10.1039/C4CS00288A |
|
(b)
doi: 10.1002/chem.v27.1 |
|
|
(c)
doi: 10.1002/tcr.v18.6 |
|
| [5] |
(a)
doi: 10.1039/D1CC05590A pmid: 33112319 |
|
(b)
doi: 10.1039/d0ob01453b pmid: 33112319 |
|
|
(c)
doi: 10.1002/asia.v11.4 pmid: 33112319 |
|
| [6] |
(a)
doi: 10.1021/ja404414q |
|
(b)
doi: 10.1021/acscatal.5b01244 |
|
|
(c)
doi: 10.1021/acs.orglett.6b01055 |
|
|
(d)
doi: 10.1039/C6RA15464F |
|
|
(e)
doi: 10.1002/anie.v56.50 |
|
|
(f)
doi: 10.1021/jacs.0c11735 |
|
| [7] |
(a)
doi: 10.1002/chem.v19.20 pmid: 34047328 |
|
(b)
doi: 10.1021/jacs.5b04858 pmid: 34047328 |
|
|
(c)
doi: 10.1039/d1ob00376c pmid: 34047328 |
|
| [8] |
(a)
doi: 10.1039/C4CC07170K pmid: 28833691 |
|
(b)
doi: 10.1021/acs.organomet.6b00530 pmid: 28833691 |
|
|
(c)
doi: 10.1021/acscatal.5b00243 pmid: 28833691 |
|
|
(d)
doi: 10.1021/acs.orglett.5b01247 pmid: 28833691 |
|
|
(e)
doi: 10.1002/chem.201703687 pmid: 28833691 |
|
|
(f)
doi: 10.1021/acscatal.9b04125 pmid: 28833691 |
|
|
(g)
doi: 10.1039/C8CC07531J pmid: 28833691 |
|
|
(h)
doi: 10.1021/acscatal.5b02540 pmid: 28833691 |
|
| [9] |
(a)
doi: 10.1039/D0CC06997C |
|
(b)
doi: 10.1038/s41467-019-13611-6 |
|
| [10] |
doi: 10.1021/acs.orglett.4c04269 |
| [11] |
doi: 10.1002/slct.v5.2 |
| [12] |
|
| [13] |
doi: 10.1021/acs.orglett.8b03015 |
| [14] |
(a)
doi: 10.1039/C8OB01182F |
|
(b)
doi: 10.1002/ajoc.v8.1 |
|
| [15] |
doi: 10.1021/acs.orglett.5b00028 |
| [16] |
(a)
doi: 10.1039/c1cs15093f pmid: 21792454 |
|
(b)
doi: 10.1002/anie.v54.27 pmid: 21792454 |
|
|
(c)
doi: 10.1021/acs.orglett.6b01746 pmid: 21792454 |
|
|
(d)
doi: 10.1039/D1CC02003J pmid: 21792454 |
|
| [17] |
doi: 10.1002/chem.v21.5 |
| [18] |
doi: 10.1039/C9CC03210J |
| [19] |
|
| [20] |
doi: 10.1021/acs.organomet.7b00004 |
| [21] |
(a)
doi: 10.1021/acs.joc.2c02157 |
|
(b)
doi: 10.1039/D3SC03861K |
|
| [22] |
doi: 10.1016/j.cclet.2024.109533 |
| [23] |
doi: 10.1021/acscatal.5c02949 |
| [24] |
doi: 10.1039/c1cc15843k |
| [25] |
(a)
doi: 10.1021/acs.orglett.7b00478 pmid: 28301167 |
|
(b)
doi: 10.1021/jo101398a pmid: 28301167 |
|
| [26] |
doi: 10.1039/C6CC06029C |
| [27] |
doi: 10.1016/S0040-4020(01)86099-3 |
| [1] | 奚晓翔, 高明, 韩立彪. 铑催化氢气还原RS—SR到硫酚/醇[J]. 有机化学, 2026, 46(3): 859-865. |
| [2] | 任才艺, 董文楠, 刘敬功, 杨爽, 房新强. 铑催化由环丙烯酮合成酰偶姻衍生物[J]. 有机化学, 2026, 46(2): 486-495. |
| [3] | 张晓锋, Aggeliki Roumana, 毛海康, 徐晶. 虎皮楠生物碱Daphniglaucin C的AB环系合成[J]. 有机化学, 2025, 45(3): 925-932. |
| [4] | 冯亚萍, 黄健, 王英杰, 孙键, 南光明, 魏伟. 可见光促进α-重氮酯参与多组分反应合成烷基二硫代磷酸酯[J]. 有机化学, 2025, 45(10): 3912-3922. |
| [5] | 王峥, 岳敏, 王宗成. 异硫氰酸酯、胺、硫鎓盐三组分反应构建S-烷基异硫脲[J]. 有机化学, 2025, 45(10): 3892-3902. |
| [6] | 杨帆, 范小梦, 姚雪婧, 米瑞杰, 于松杰, 李兴伟, 肖建. 铑(III)催化氧化锍叶立德和重氮化合物环化偶联反应[J]. 有机化学, 2025, 45(1): 331-342. |
| [7] | 文思, 丁宇浩, 田青于, 葛进, 程国林. 铑(III)催化苯甲亚胺酸乙酯和CF3-亚胺氧锍叶立德C—H 活化/环化反应合成CF3-1H-苯并[de][1,8]萘吡啶[J]. 有机化学, 2024, 44(1): 291-300. |
| [8] | 张彦波, 孙萌. 铑催化碳酸亚乙烯酯与吲哚啉C(7)位C—H甲酰甲基化反应[J]. 有机化学, 2023, 43(8): 2905-2912. |
| [9] | 汤振, 皮超, 吴养洁, 崔秀灵. 铑催化2-芳基-2H-吲唑与硫叶立德的酰甲基化/串联环化反应高效构建6-芳基吲唑并[2,3-a]喹啉类衍生物[J]. 有机化学, 2023, 43(3): 1187-1196. |
| [10] | 李硕, 王明亮, 周来运, 王兰芝. 磁性纳米负载对甲苯磺酸催化串联合成稠合多环的1,5-苯并氧氮杂䓬类化合物[J]. 有机化学, 2023, 43(11): 3977-3988. |
| [11] | 刘晓洁, 徐必平, 苏伟平. 铑催化羧酸原位生成酰氟的脱羰Suzuki-Miyaura偶联[J]. 有机化学, 2022, 42(7): 2184-2191. |
| [12] | 张玉荣, 王晗, 茆勇军, 施世良. 镍催化丁二烯、亚胺和烯基硼酸的三组分偶联反应[J]. 有机化学, 2022, 42(4): 1198-1209. |
| [13] | 肖立伟, 刘光仙, 任萍, 吴彤桐, 卢玉伟, 孔洁. 单质硫: 合成含硫杂环的优质硫源[J]. 有机化学, 2022, 42(4): 1002-1012. |
| [14] | 穆思宇, 李红霞, 伍智林, 彭俊梅, 陈锦杨, 何卫民. 电催化肼、丙二酮和2-溴丙二酸二乙酯三组分合成4-溴吡唑[J]. 有机化学, 2022, 42(12): 4292-4299. |
| [15] | 王家状, 滕丽果, 熊绍棋, 肖铁波, 江玉波. Rh催化N-磺酰腙的偕-二氟烯丙基化反应[J]. 有机化学, 2022, 42(11): 3658-3667. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||